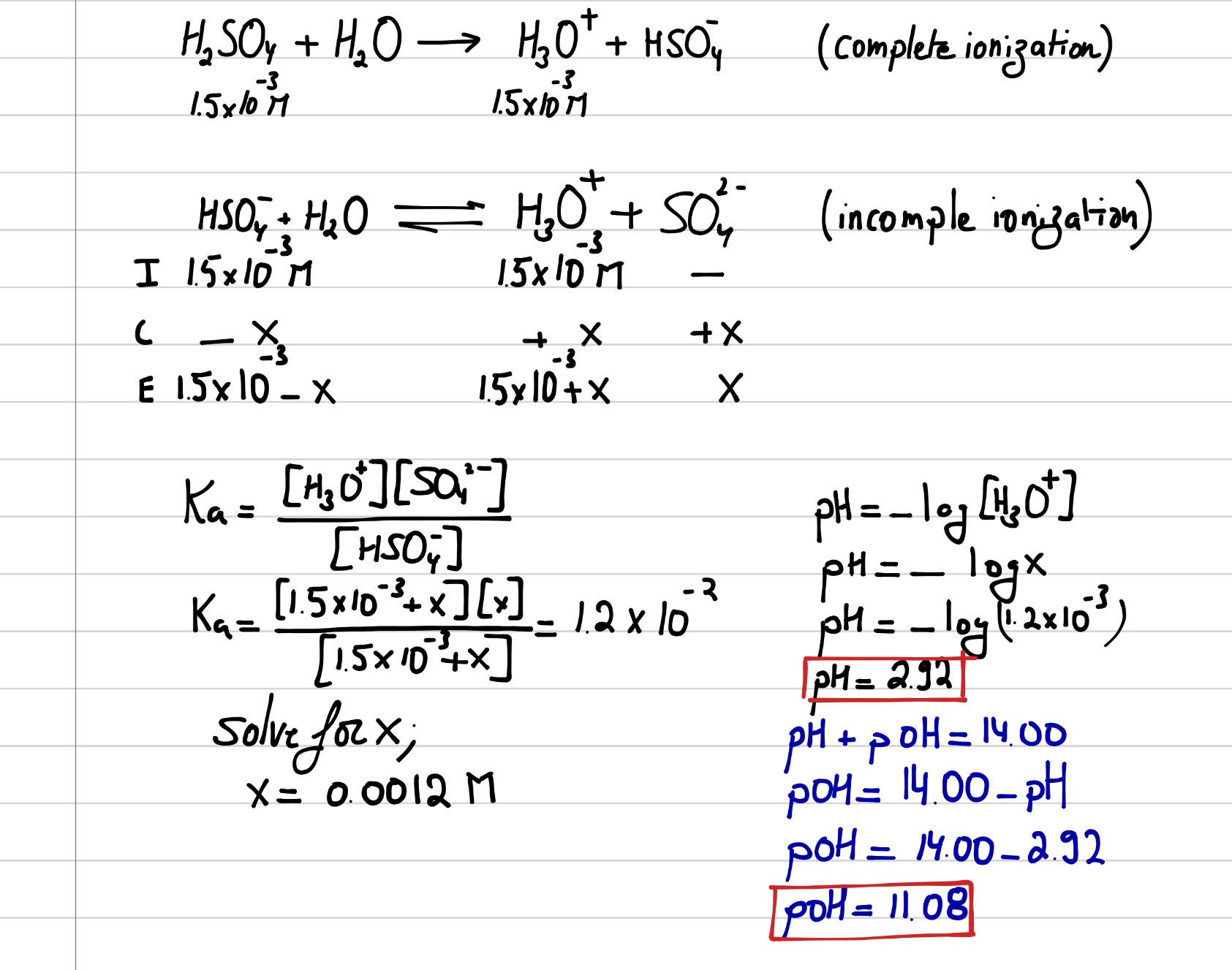SOLVED: 8) Sulfuric acid is a strong acid, and the pKa 2 of HSO - 4 is 1.92. What is the pH of a 0.100 M NaHSO 4 solution? 9) What is
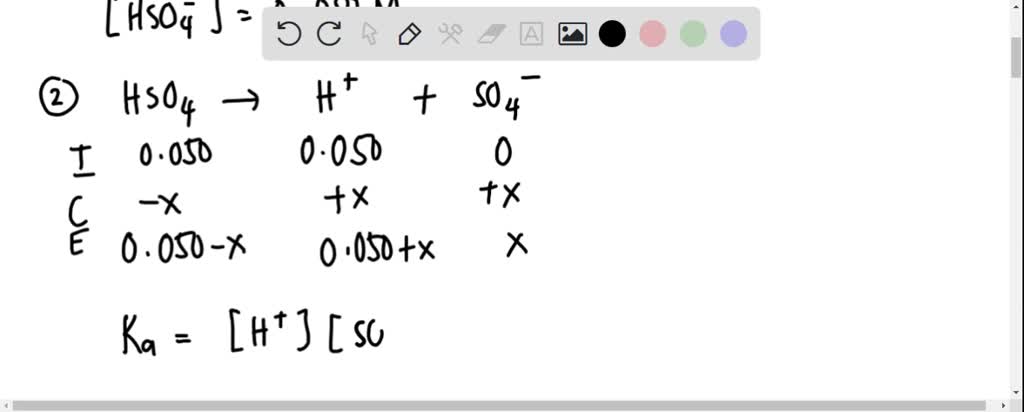
SOLVED: For sulfuric acid, Ka1 > 1, and Ka2 is 0.012. Calculate the pH of a 1.2 M solution of H2SO4 . (Steps would be helpful, thank you!)

Dissociation Constants of Perchloric and Sulfuric Acids in Aqueous Solution | The Journal of Physical Chemistry B

If Ka1 and Ka2 of sulphuric acid are 1 × 10^-2 and 1 × 10^-6 respectively, then concentration of sulphate ions in 0.01 MH2SO4 solution will be:

How much heat does 240 millilitres sulfuric acid make when 240 millilitres of water is added? - Quora

Ammonia Catalyzed Formation of Sulfuric Acid in Troposphere: The Curious Case of a Base Promoting Acid Rain | The Journal of Physical Chemistry A
![SOLVED: Example 14.13 What is the pH of a 0.0025 M sulfuric acid solution? What is [S042-]? HzSO4 (aq) + Hz0 () H:O+ (aq) + HSO4 (aq) HSOz (aq) + HzO () SOLVED: Example 14.13 What is the pH of a 0.0025 M sulfuric acid solution? What is [S042-]? HzSO4 (aq) + Hz0 () H:O+ (aq) + HSO4 (aq) HSOz (aq) + HzO ()](https://cdn.numerade.com/ask_images/0c1eac2937604c57b404c3a0e47eb149.jpg)
SOLVED: Example 14.13 What is the pH of a 0.0025 M sulfuric acid solution? What is [S042-]? HzSO4 (aq) + Hz0 () H:O+ (aq) + HSO4 (aq) HSOz (aq) + HzO ()