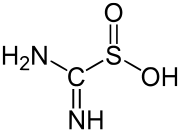
Profiling the reactivity of cyclic C-nucleophiles towards electrophilic sulfur in cysteine sulfenic acid - Chemical Science (RSC Publishing) DOI:10.1039/C5SC02569A
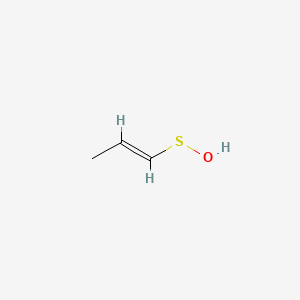
Molecules | Free Full-Text | Transient Sulfenic Acids in the Synthesis of Biologically Relevant Products

Mechanistic studies of a novel C-S lyase in ergothioneine biosynthesis: the involvement of a sulfenic acid intermediate | Scientific Reports

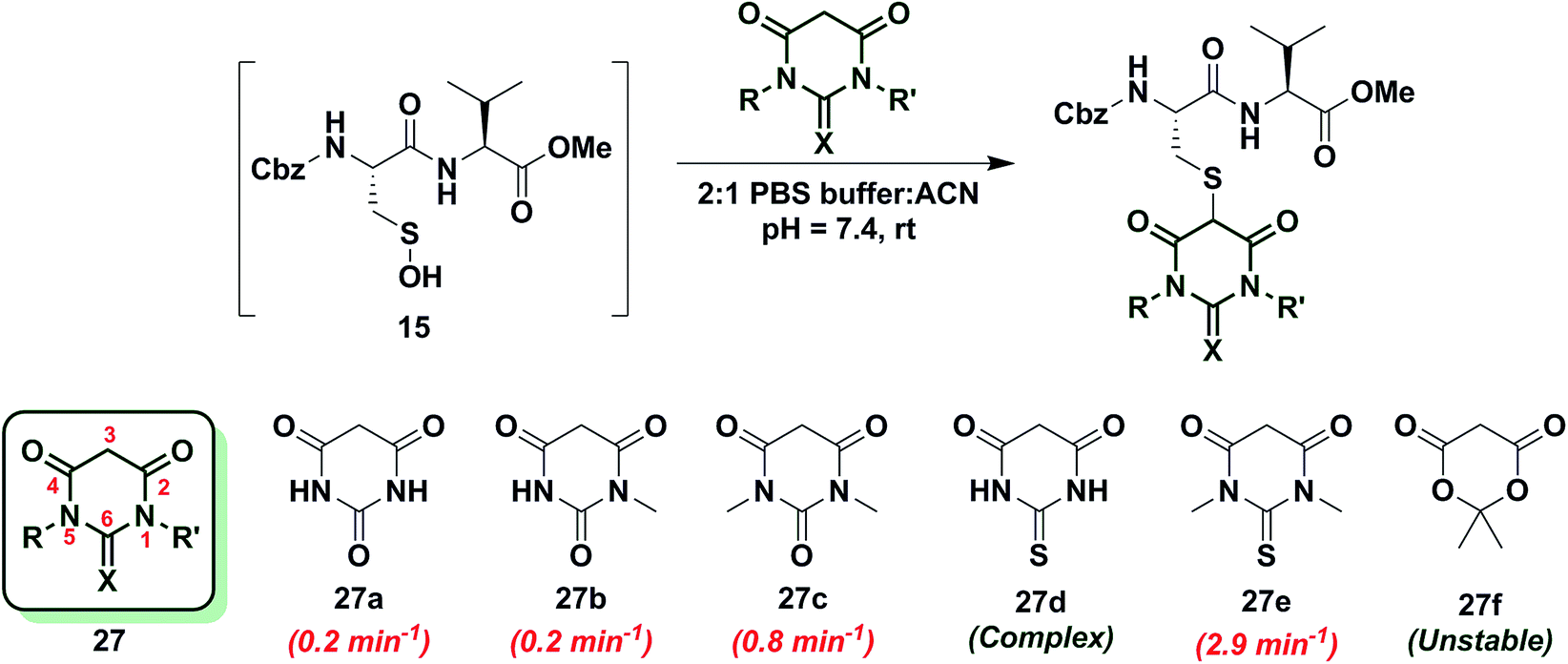
a) A dipeptide-based sulfenic acid model. (b) Various 1,3-diketones... | Download Scientific Diagram

Mining the Thiol Proteome for Sulfenic Acid Modifications Reveals New Targets for Oxidation in Cells | ACS Chemical Biology
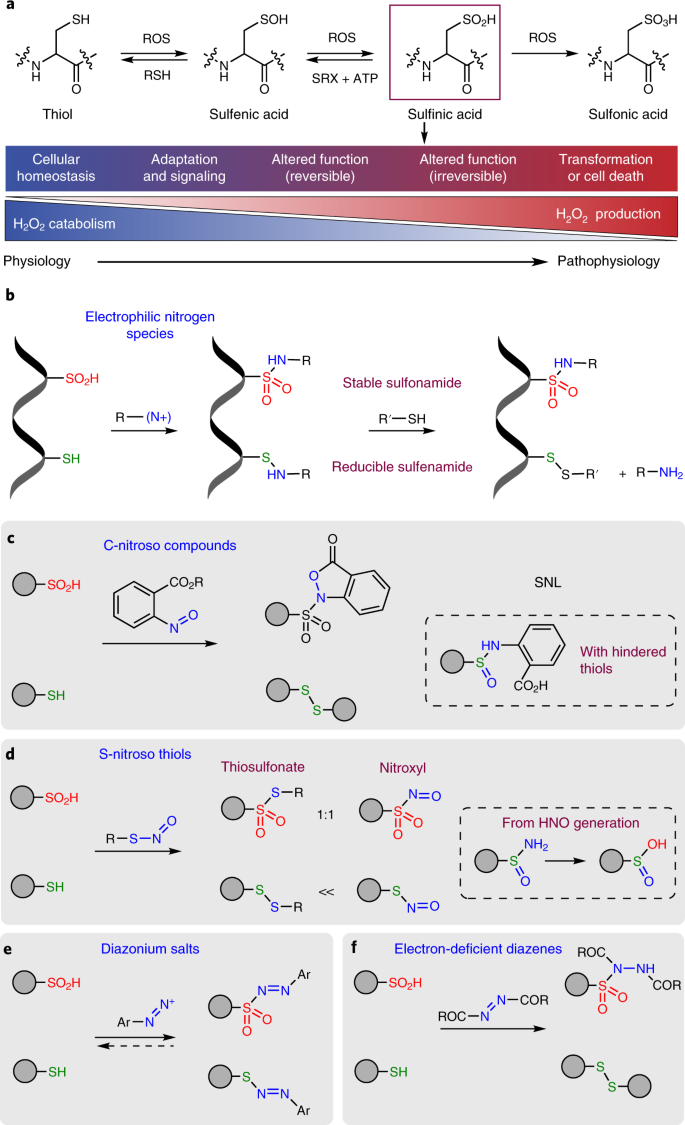
Chemical proteomics reveals new targets of cysteine sulfinic acid reductase | Nature Chemical Biology

Cysteine sulfenic Acid as an Intermediate in Disulfide Bond Formation and Nonenzymatic Protein Folding | Biochemistry






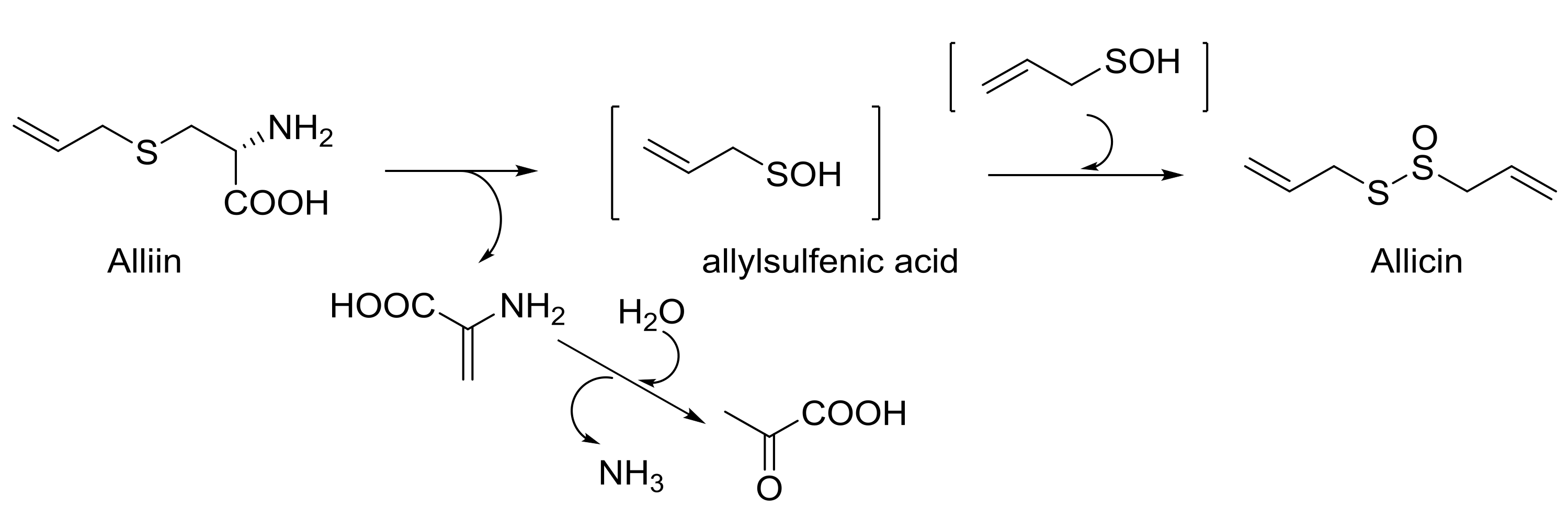
.jpg-650.jpg)