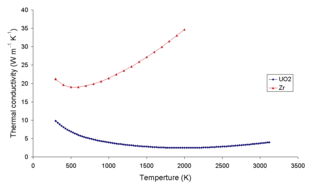
Suppressed thermal conductivity in hyperstoichiometric uranium dioxide controlled by phonon lifetimes: Applied Physics Letters: Vol 121, No 1

Specific heat capacity vs. temperature for UO2. The data in this work... | Download Scientific Diagram
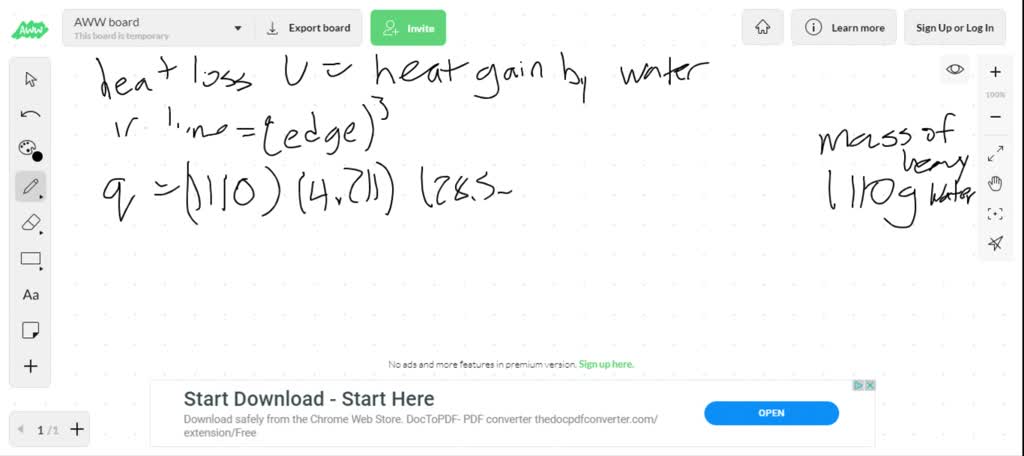
SOLVED:A cubic piece of uranium metal (specific heat capacity = 0.117 J /^∘ C ·g at 200.0^∘ C is dropped into 1.00 L deuterium oxide ("heavy water," specific heat capacity =4.211 J /
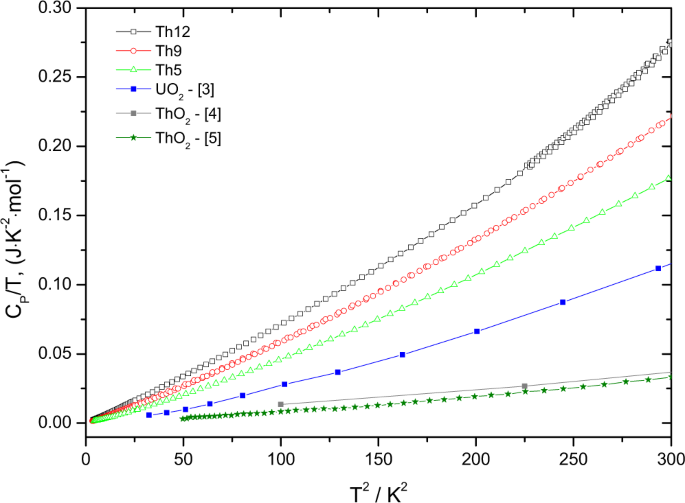
The effect of lattice disorder on the low-temperature heat capacity of (U1−yThy)O2 and 238Pu-doped UO2 | Scientific Reports

Specific heat capacity vs. temperature for UO2. The data in this work... | Download Scientific Diagram

Specific heat as a function of temperature for pure urania and Beryllia... | Download Scientific Diagram

SOLVED: A cubic piece of uranium metal (specific heat capacity =0.117 J /^∘C·g ) at 200.0^∘C is dropped into 1.00 L deuterium oxide ("heavy water," specific heat capacity =4.211 J /^∘C·g )
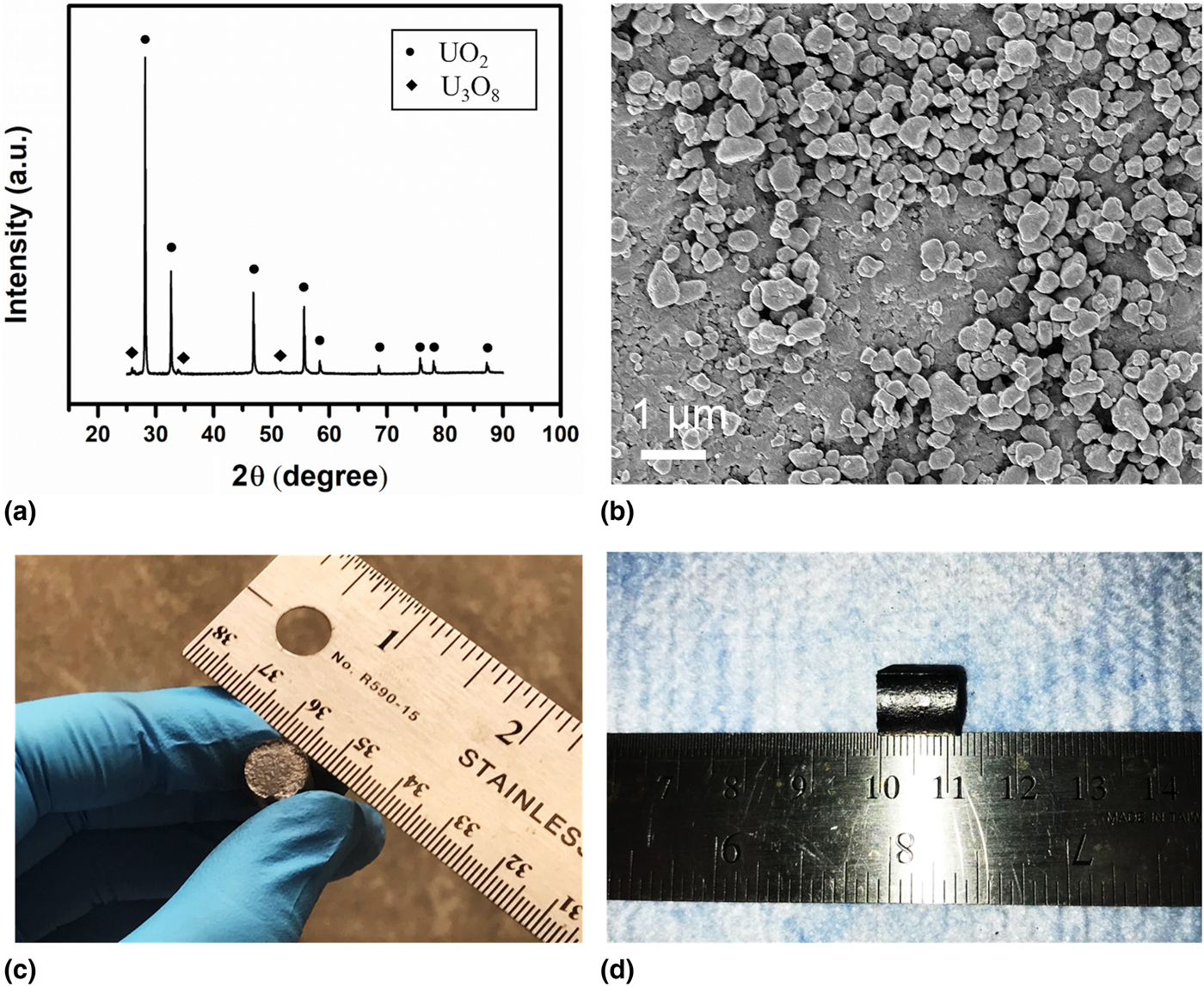
Consolidation of commercial-size UO2 fuel pellets using spark plasma sintering and microstructure/microchemical analysis | MRS Communications | Cambridge Core