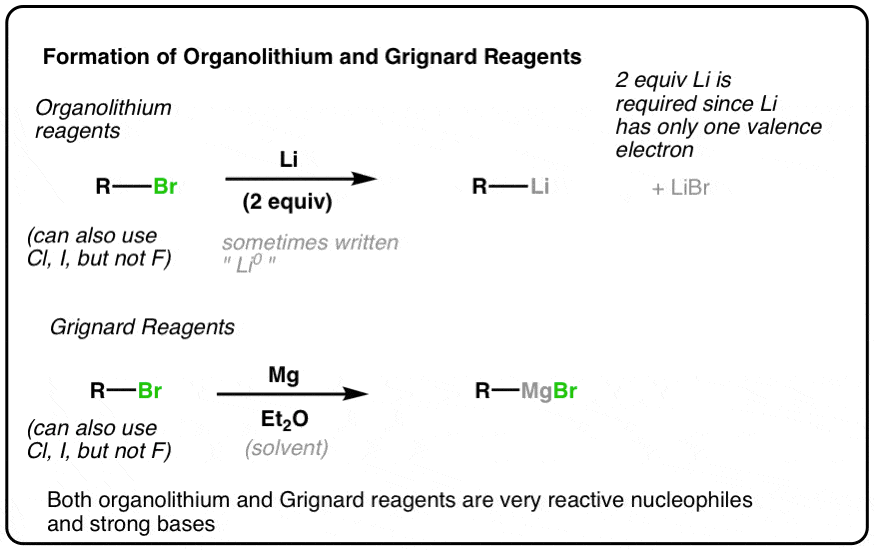
Selective C–P(O) Bond Cleavage of Organophosphine Oxides by Sodium | The Journal of Organic Chemistry

Enantioselective Cu-Catalyzed Arylation of Secondary Phosphine Oxides with Diaryliodonium Salts toward the Synthesis of P-Chiral Phosphines | Semantic Scholar

Mechanistic aspects of the stereospecific reduction of chiral hydroxyalkyl phosphinates and phosphine oxides
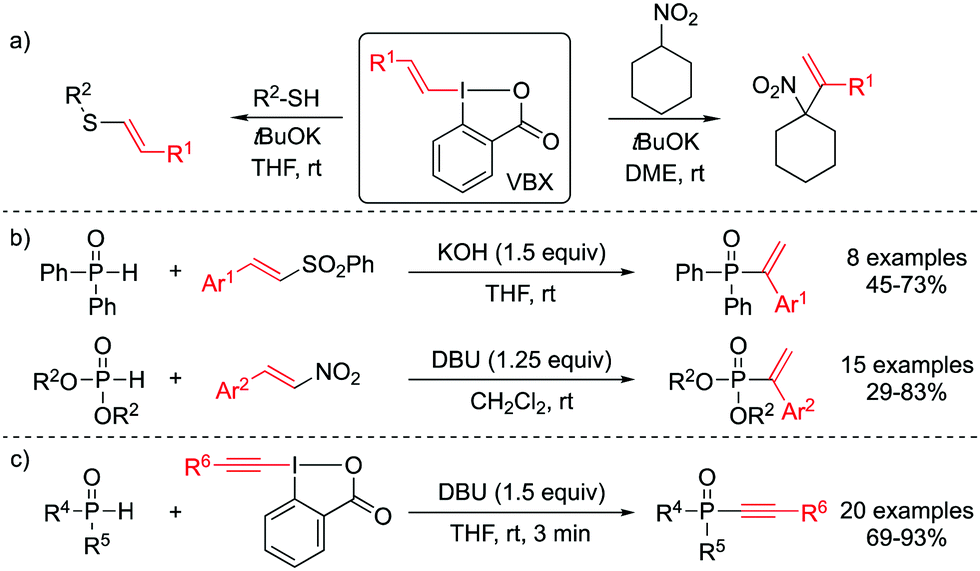
Transition metal-free and regioselective vinylation of phosphine oxides and H -phosphinates with VBX reagents - Chemical Communications (RSC Publishing) DOI:10.1039/D0CC05992G

Consecutive dynamic resolutions of phosphine oxides - Chemical Science (RSC Publishing) DOI:10.1039/C3SC52913D

Ni-Catalyzed Asymmetric Allylation of Secondary Phosphine Oxides | Journal of the American Chemical Society

Ni-Catalyzed Asymmetric Allylation of Secondary Phosphine Oxides | Journal of the American Chemical Society
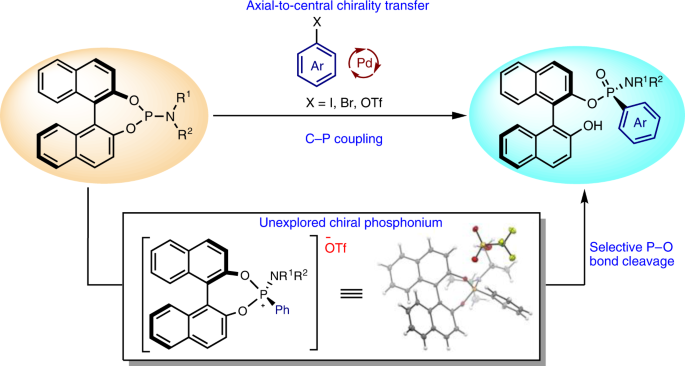
P-chirogenic phosphorus compounds by stereoselective Pd-catalysed arylation of phosphoramidites | Nature Catalysis

C−As Bond Formation Reactions for the Preparation of Organoarsenic(III) Compounds - Tay - 2020 - Chemistry – An Asian Journal - Wiley Online Library

The First Phosphine Oxide Ligand Precursors for Transition Metal Catalyzed Cross-Coupling Reactions: C-C, C-N, and C-S Bond Formation on Unactivated Aryl Chlorides
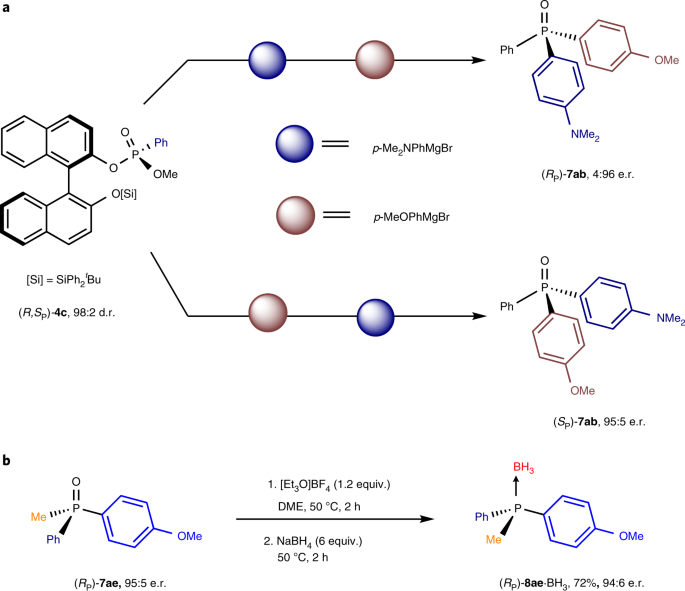
P-chirogenic phosphorus compounds by stereoselective Pd-catalysed arylation of phosphoramidites | Nature Catalysis

Secondary phosphine oxides: Versatile ligands in transition metal-catalyzed cross-coupling reactions - ScienceDirect

Mechanistic aspects of the stereospecific reduction of chiral hydroxyalkyl phosphinates and phosphine oxides

Efficient Asymmetric Synthesis of P-Chiral Phosphine Oxides via Properly Designed and Activated Benzoxazaphosphinine-2-oxide Agents | Journal of the American Chemical Society

Secondary Phosphine Oxide Functionalized Gold Clusters and Their Application in Photoelectrocatalytic Hydrogenation Reactions | Journal of the American Chemical Society

Facial conversion of secondary phosphine oxides R1R2P(O)H to chlorophosphines R1R2PCl by acetyl chloride - ScienceDirect

Enantioselective Cu-Catalyzed Arylation of Secondary Phosphine Oxides with Diaryliodonium Salts toward the Synthesis of P-Chiral Phosphines | Semantic Scholar

Enantioselective Cu-Catalyzed Arylation of Secondary Phosphine Oxides with Diaryliodonium Salts toward the Synthesis of P-Chiral Phosphines | Semantic Scholar