
Effect of Ethylene Oxide and Gamma (γ-) Sterilization on the Properties of a PLCL Polymer Material in Balloon Implants | ACS Omega

Controls of Fugitive Emissions of Residual Ethylene Oxide inCommercial Medical-supply Sterilization | Semantic Scholar
UNE EN ISO 10993-7:2009 Biological evaluation of medical devices - Part 7: Ethylene oxide sterilization residuals (ISO 10993-7:2008) - European Standards

Re-validation of EO Sterilization - When is re-validation required? - Medical Device Academy Medical Device Academy
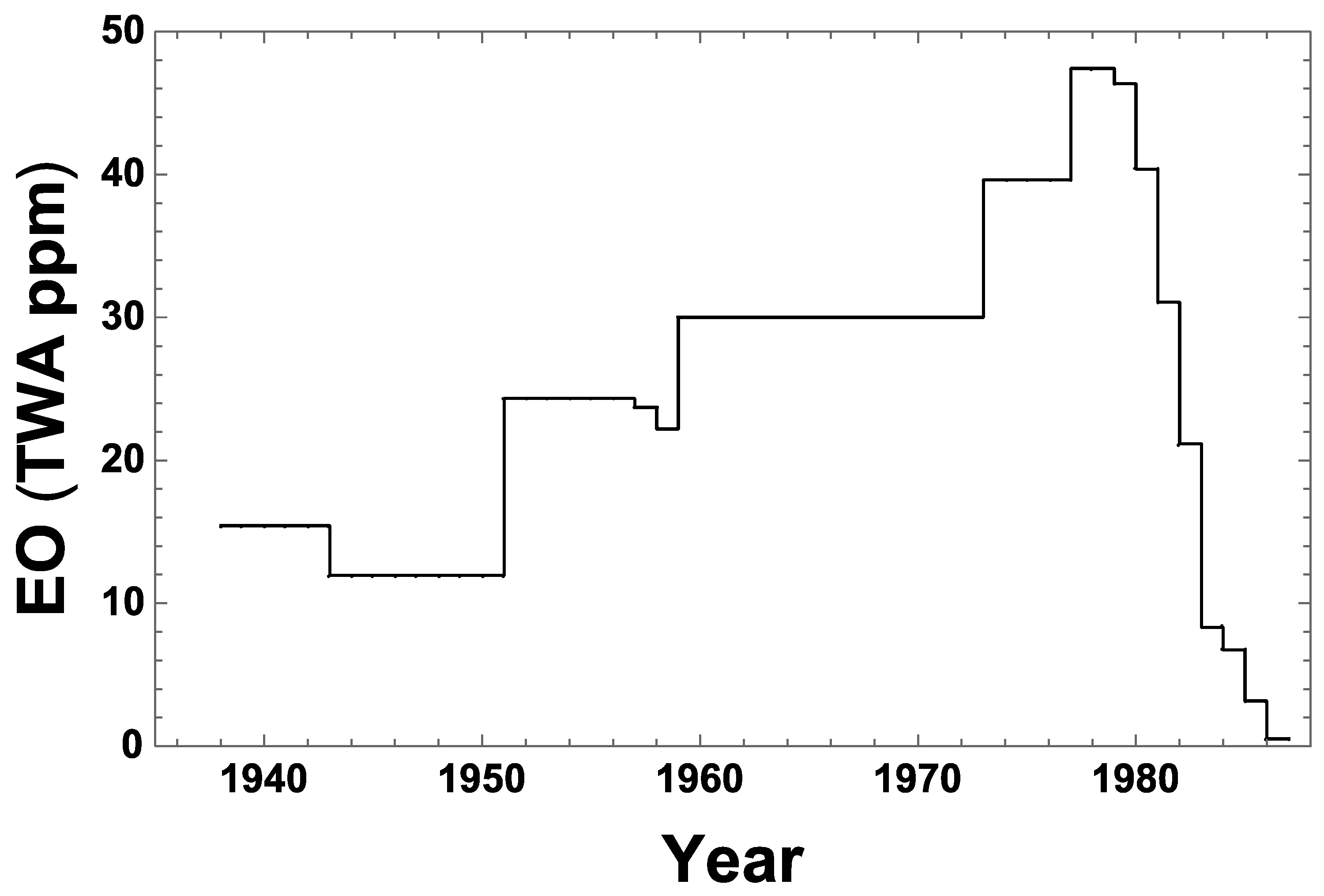
IJERPH | Free Full-Text | Reevaluation of Historical Exposures to Ethylene Oxide Among U.S. Sterilization Workers in the National Institute of Occupational Safety and Health (NIOSH) Study Cohort

AAMI TIR19:1998 -- Guidance for ANSI/AAMI/ISO 10993-7:1995, Biological evaluation of medical devices - Part 7: Ethylene oxide sterilization residuals: AAMI: Amazon.com: Books
Biocompatibility & analysis of medical devices according to ISO 10993 - ISO 10993-7: Ethylene oxide sterilization residuals - Services - Danish Technological Institute

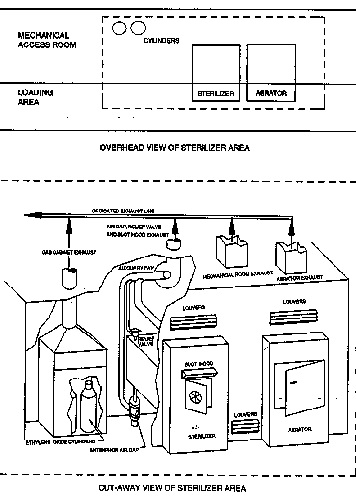

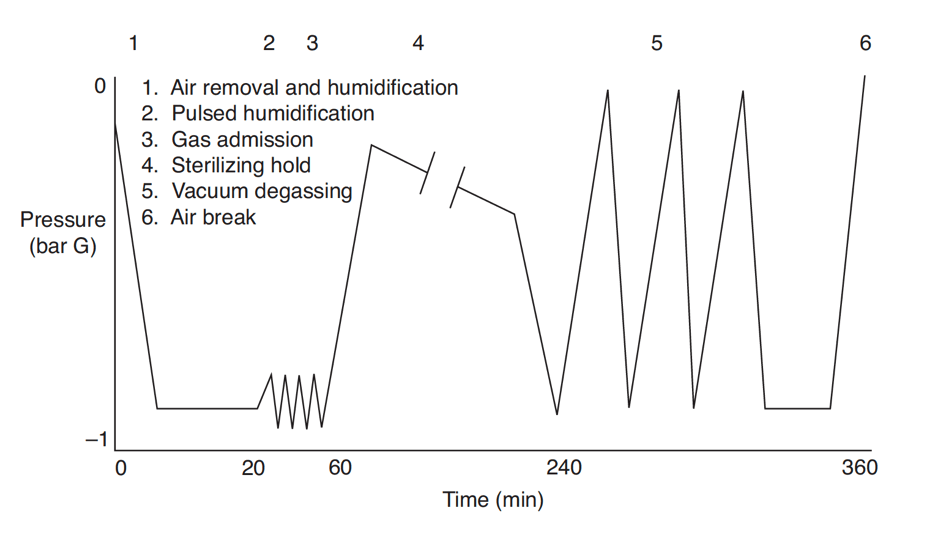







![PDF] Ethylene Oxide Gas Sterilization of Medical Devices. | Semantic Scholar PDF] Ethylene Oxide Gas Sterilization of Medical Devices. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/9be707654f44177a7faaf9f2cbf7bb13c2464b95/7-Figure5-1.png)





