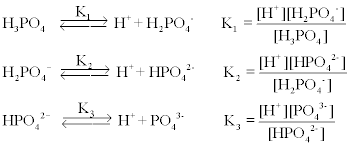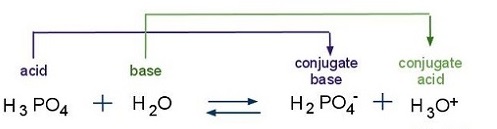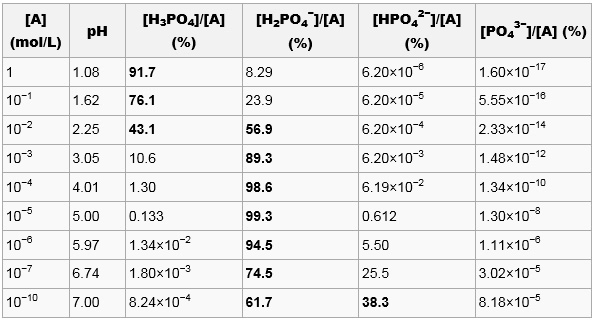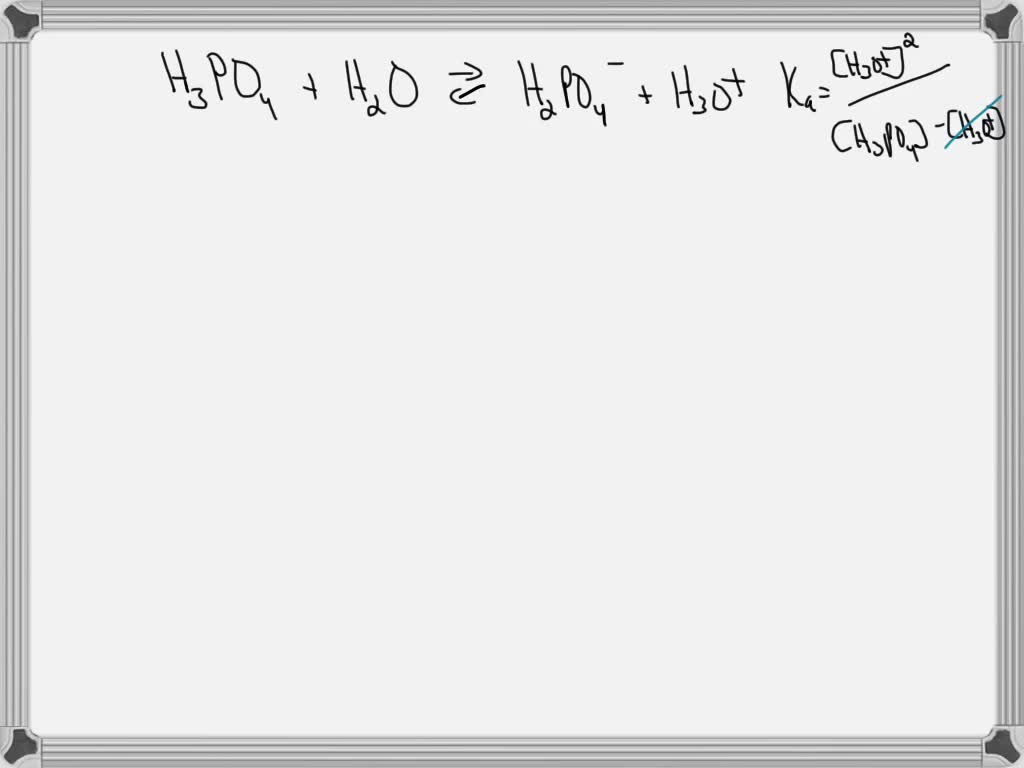
SOLVED: The acid-dissociation constants of phosphoric acid, H3PO4, are Ka1 = 7.5 × 10-3 , Ka2 = 6.2 × 10-8 and Ka3 = 4.2 × 10-14 at 25.0°C. What is the pH of a 7.40 M aqueous solution of phosphoric acid?.
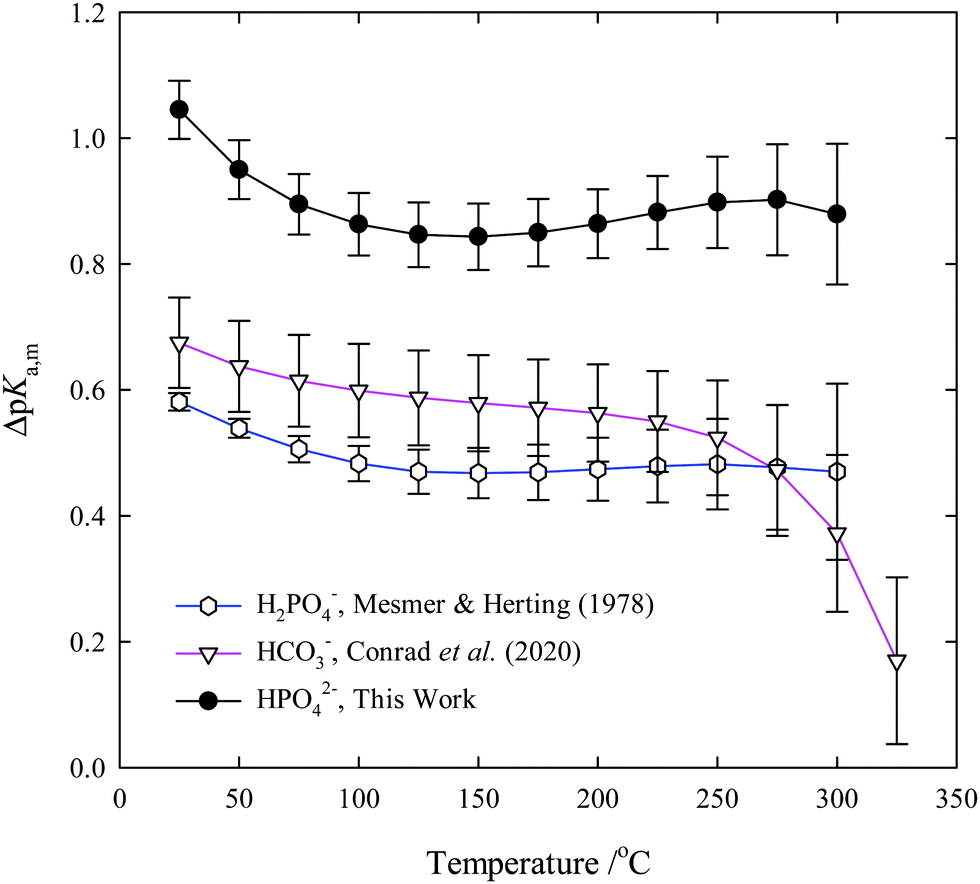
Third dissociation constant of phosphoric acid in H 2 O and D 2 O from 75 to 300 °C at p = 20.4 MPa using Raman spectroscopy and a titanium-sapphire f ... -

Phosphorous acid Acetic acid Phosphoric acid Acid dissociation constant, salt, angle, text, logo png | PNGWing

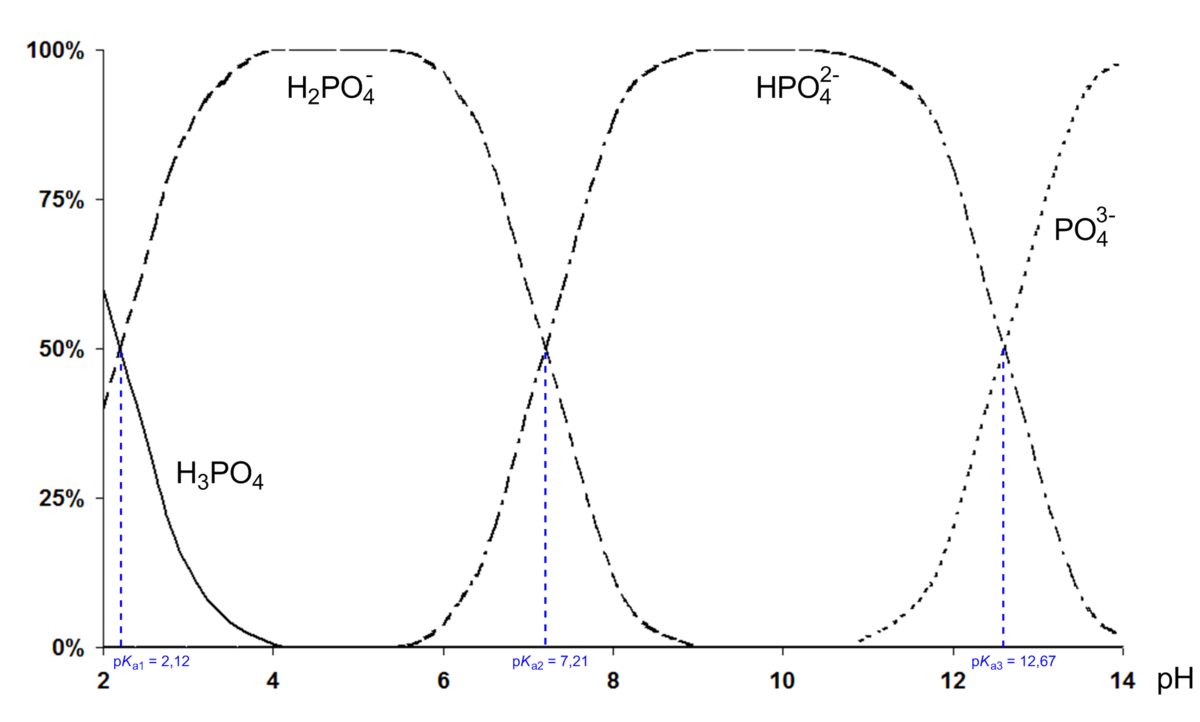
SOLVED: Phosphoric acid, H3PO4 is a triprotic acid. pKa1= 2.16 pKa2=7.21 pKa3=12.32 Write three separate acid dissociation reactions for the three acidic protons. Make sure to indicate which Ka and Kb value
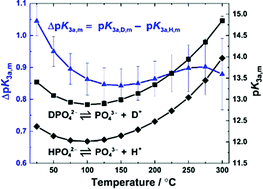
Third dissociation constant of phosphoric acid in H2O and D2O from 75 to 300 °C at p = 20.4 MPa using Raman spectroscopy and a titanium-sapphire flow cell - Physical Chemistry Chemical Physics (RSC Publishing)