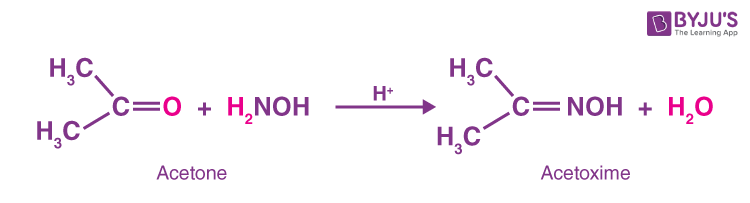
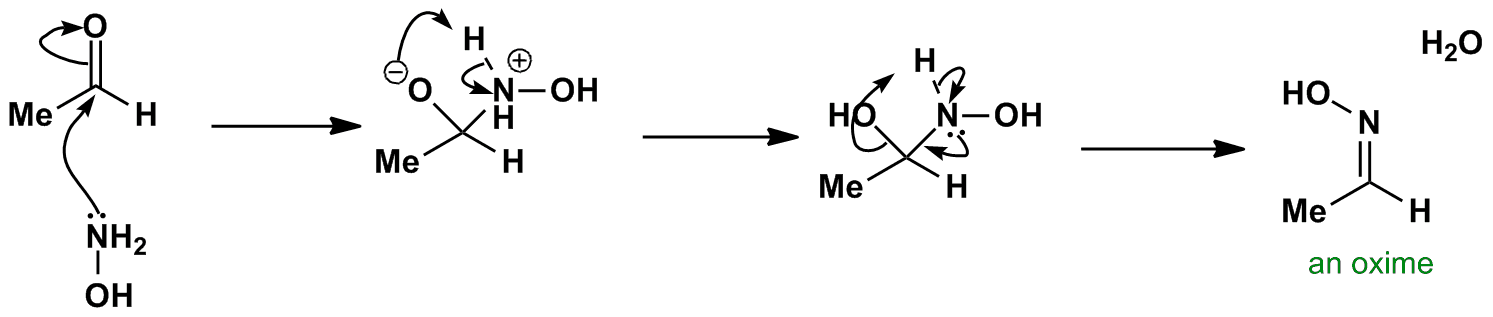
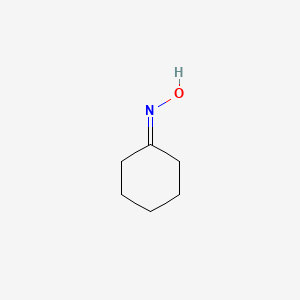
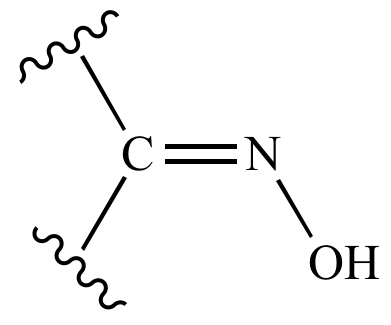
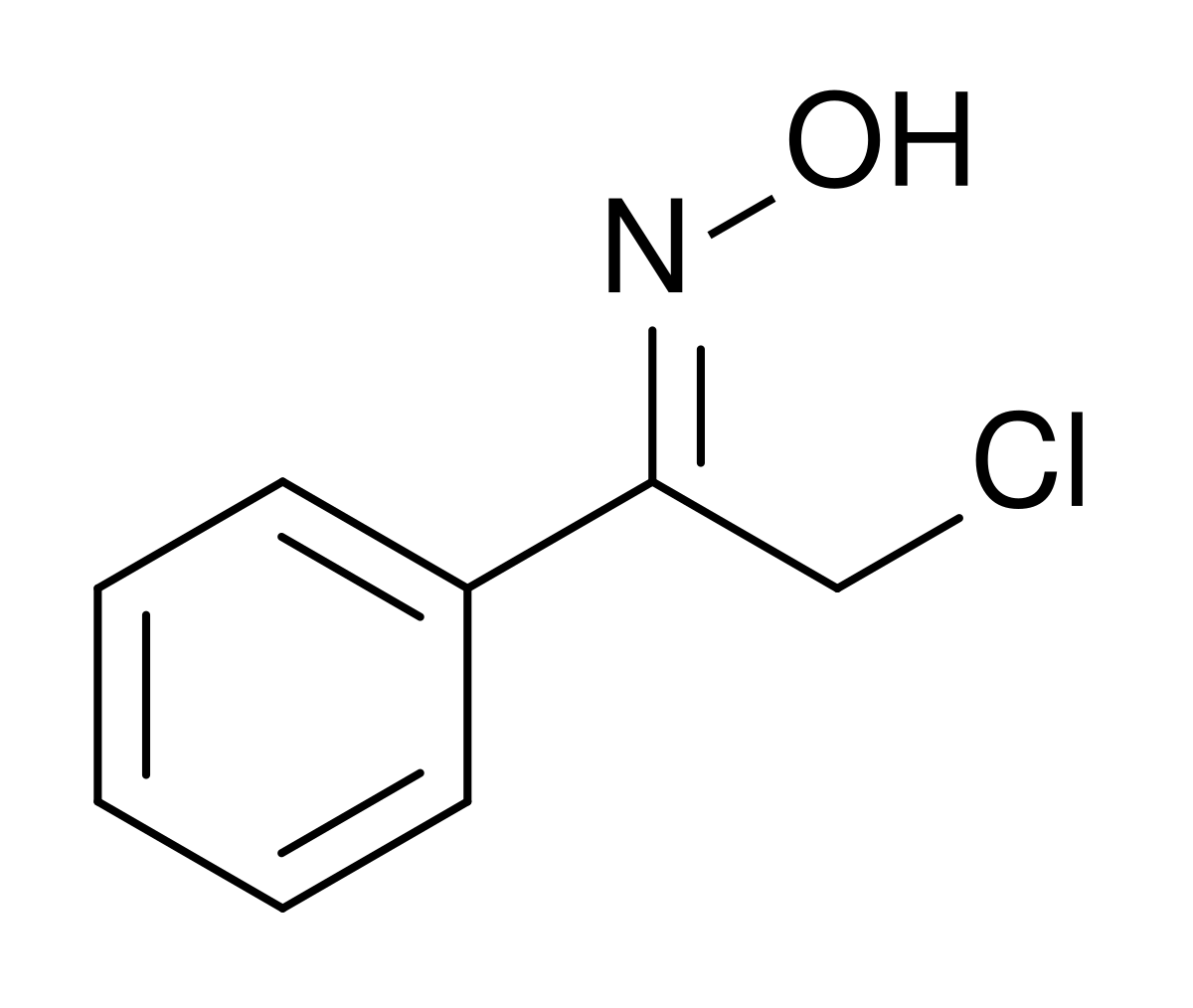
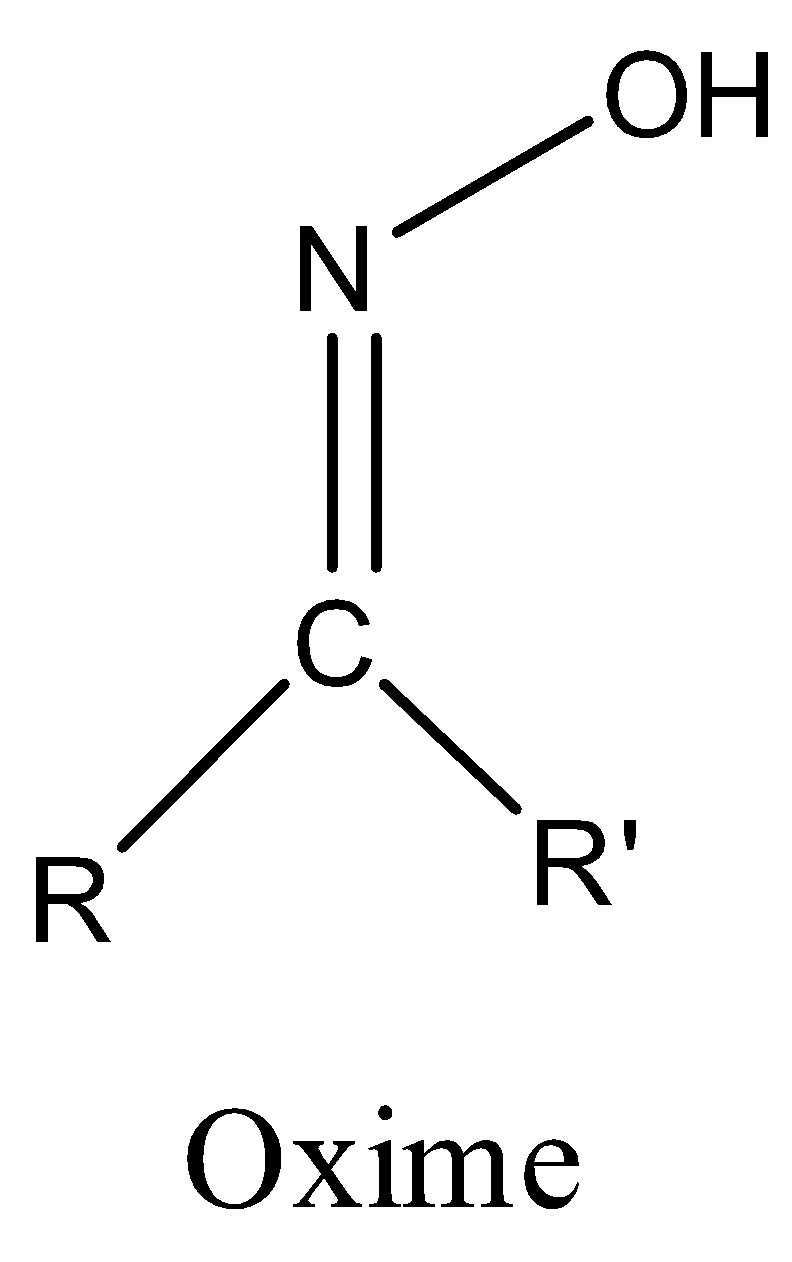
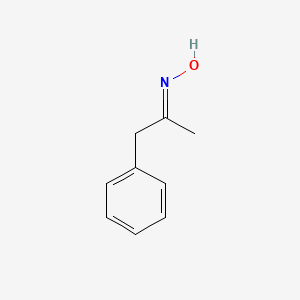
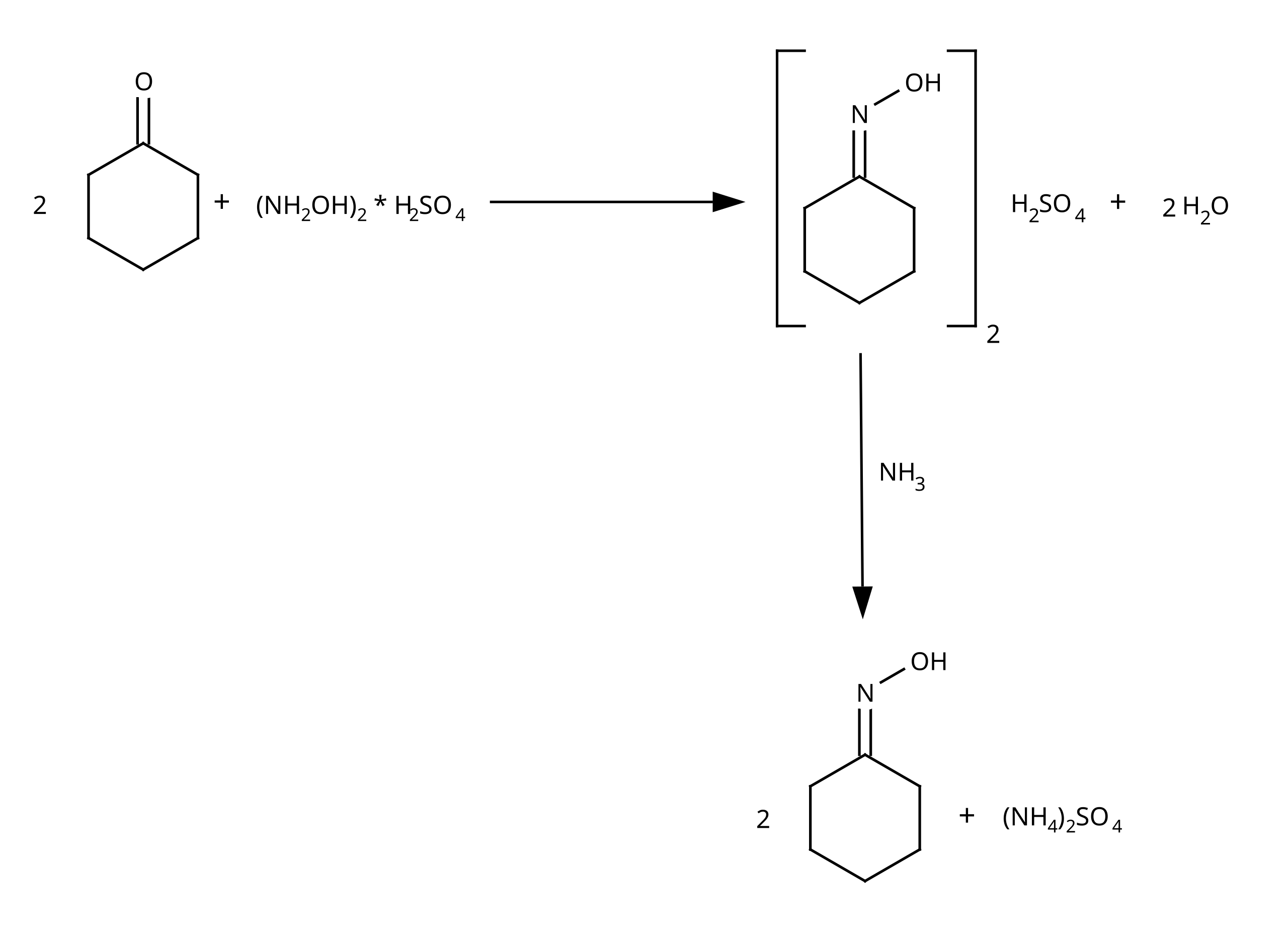
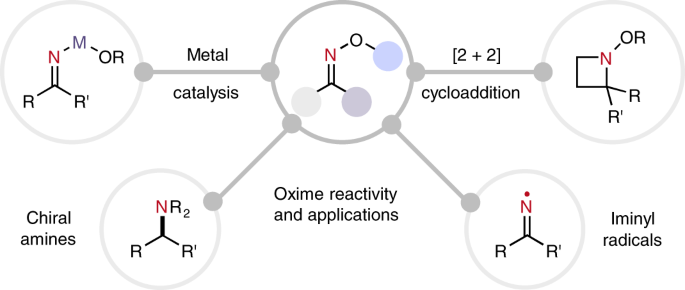
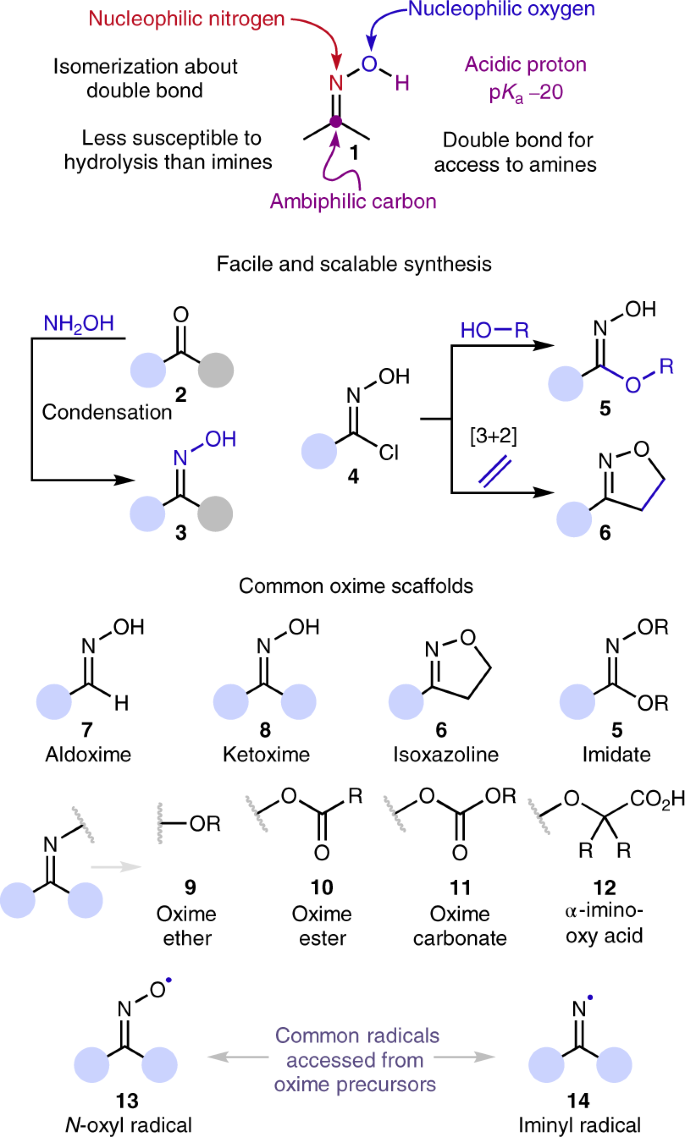
Oxime-based linker libraries as a general approach for the rapid generation and screening of multidentate inhibitors | Nature Protocols
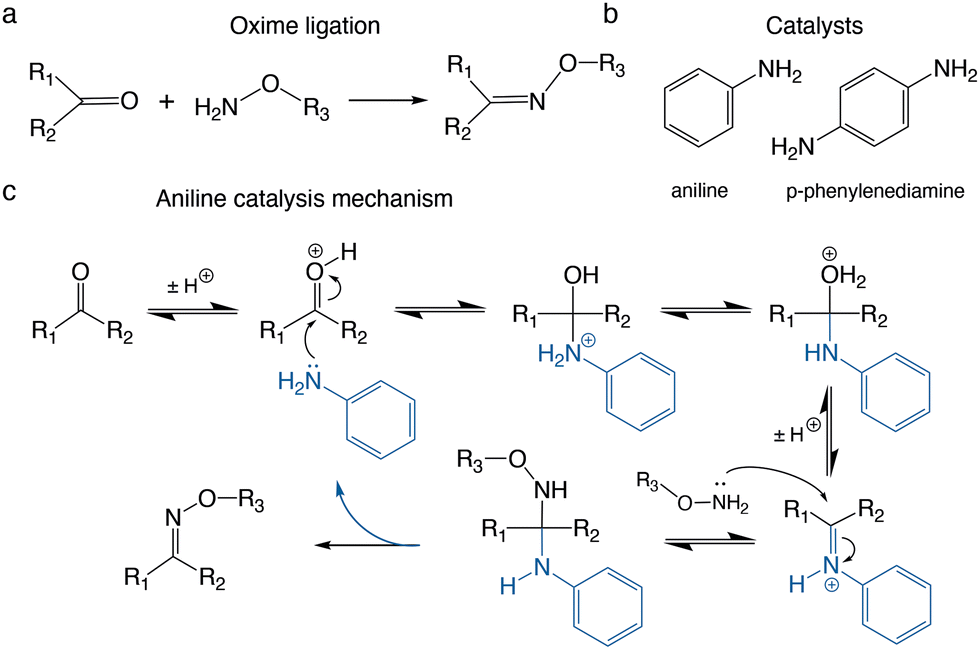
Expanding the versatility and scope of the oxime ligation: rapid bioconjugation to disulfide-rich peptides - Chemical Communications (RSC Publishing) DOI:10.1039/D2CC03752A
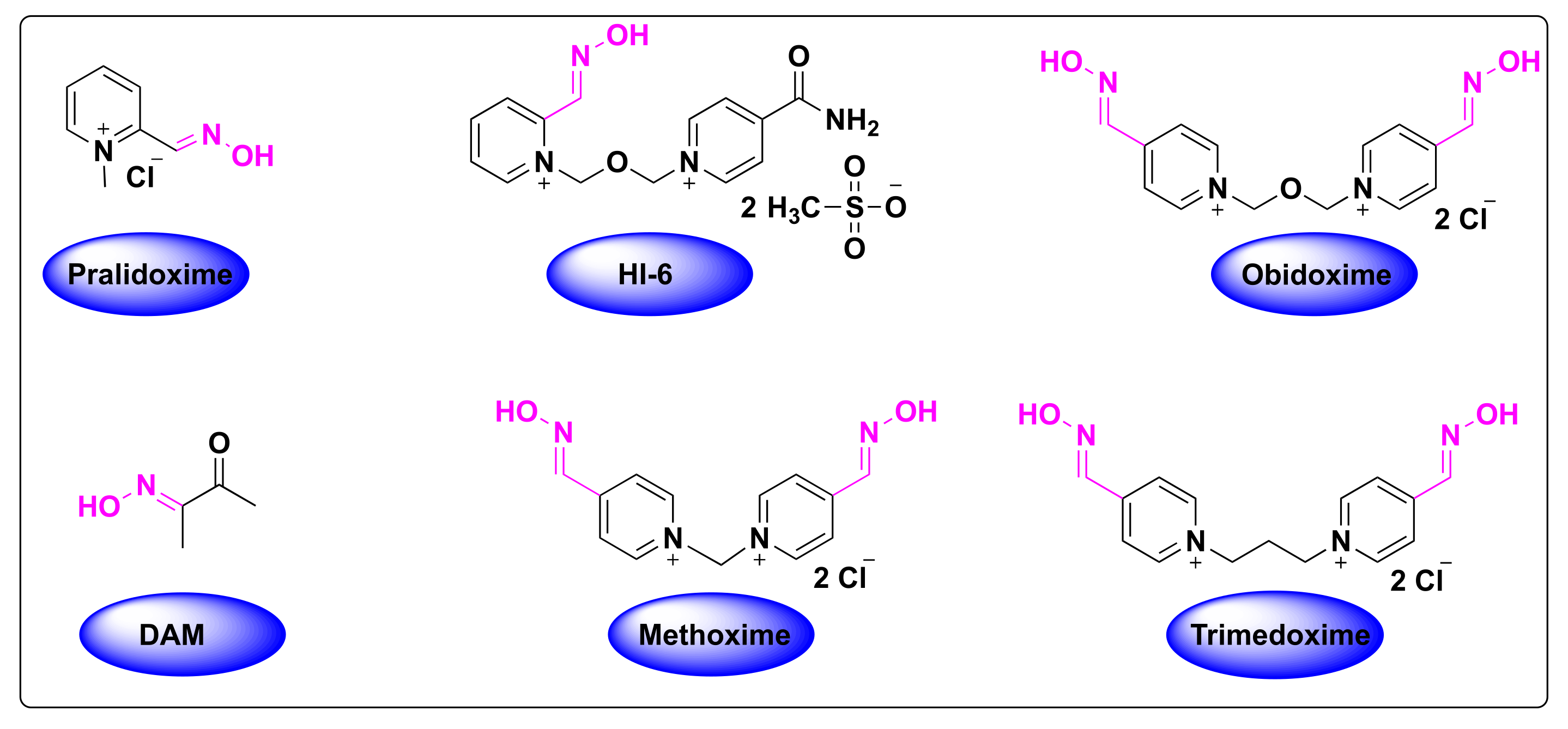
Pharmaceuticals | Free Full-Text | FDA-Approved Oximes and Their Significance in Medicinal Chemistry
![Report: [3,3]-Rearrangements of O-Vinyl Oximes: Stereoselective Synthesis of 1,4-Dicarbonyl Compounds (56th Annual Report on Research Under Sponsorship of The American Chemical Society Petroleum Research Fund) Report: [3,3]-Rearrangements of O-Vinyl Oximes: Stereoselective Synthesis of 1,4-Dicarbonyl Compounds (56th Annual Report on Research Under Sponsorship of The American Chemical Society Petroleum Research Fund)](https://acswebcontent.acs.org/prfar/2011/images/Paper_11567_abstract_15187_0.gif)