Copper-Catalyzed Asymmetric Addition of Tertiary Carbon Nucleophiles to 2H-Azirines: Access to Chiral Aziridines with Vicinal Tetrasubstituted Stereocenters | Organic Letters

Control over Ligand-Exchange Positions of Thiolate-Protected Gold Nanoclusters Using Steric Repulsion of Protecting Ligands | Journal of the American Chemical Society

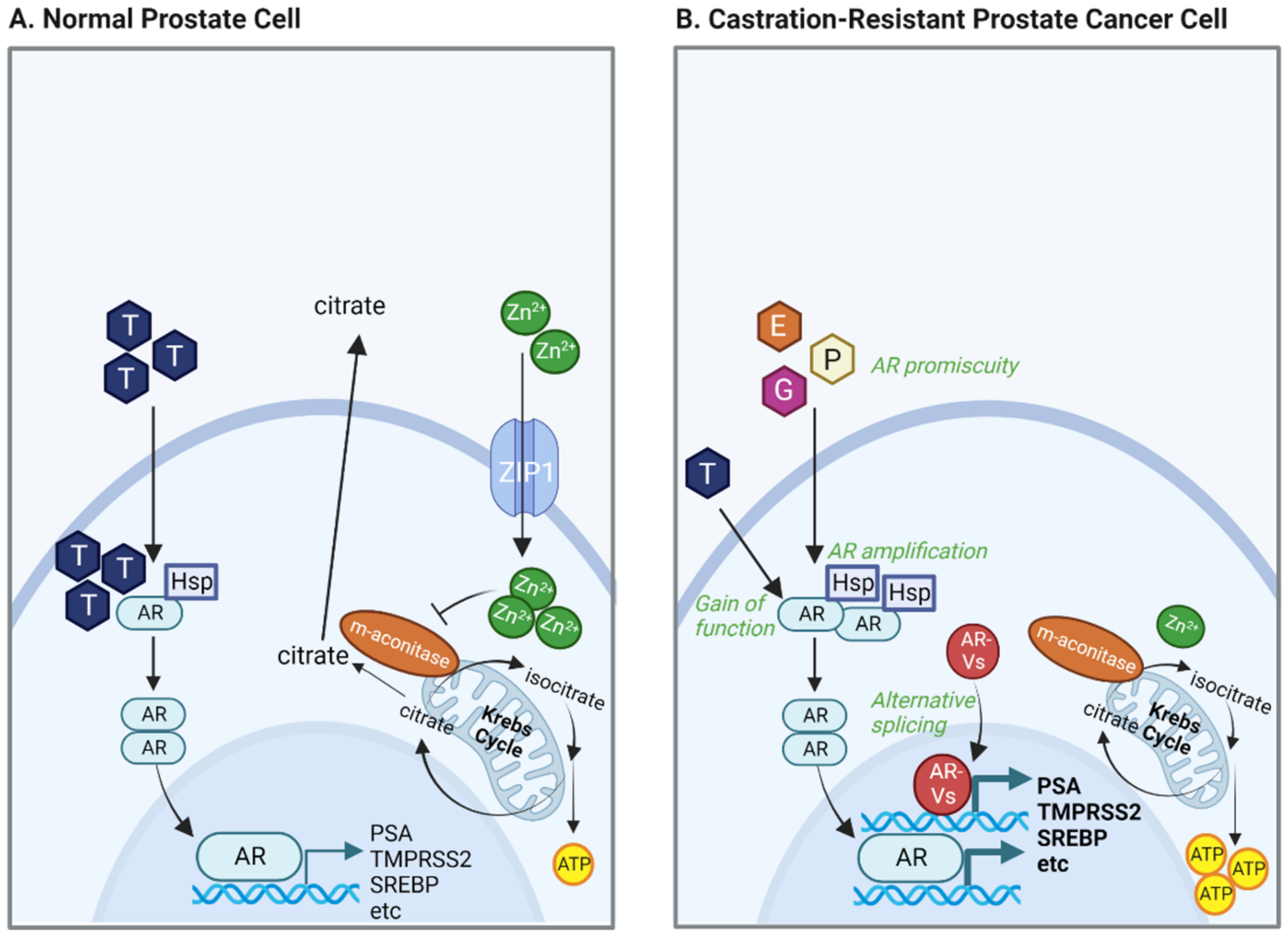
Development of Protein Degradation Inducers of Androgen Receptor by Conjugation of Androgen Receptor Ligands and Inhibitor of Apoptosis Protein Ligands | Journal of Medicinal Chemistry

Stereoselective Synthetic Strategies to (−)‐Cannabidiol - Maiocchi - 2022 - ChemistrySelect - Wiley Online Library

Palladium-catalyzed oxidative arene C–H alkenylation reactions involving olefins: Trends in Chemistry

Explanation for Main Features of Structure–Genotoxicity Relationships of Aromatic Amines by Theoretical Studies of Their Activation Pathways in CYP1A2 | Journal of the American Chemical Society

Electrospun Lignin-Derived Carbon Micro- and Nanofibers: A Review on Precursors, Properties, and Applications | ACS Sustainable Chemistry & Engineering

CO2-Promoted Reactions: An Emerging Concept for the Synthesis of Fine Chemicals and Pharmaceuticals | ACS Catalysis
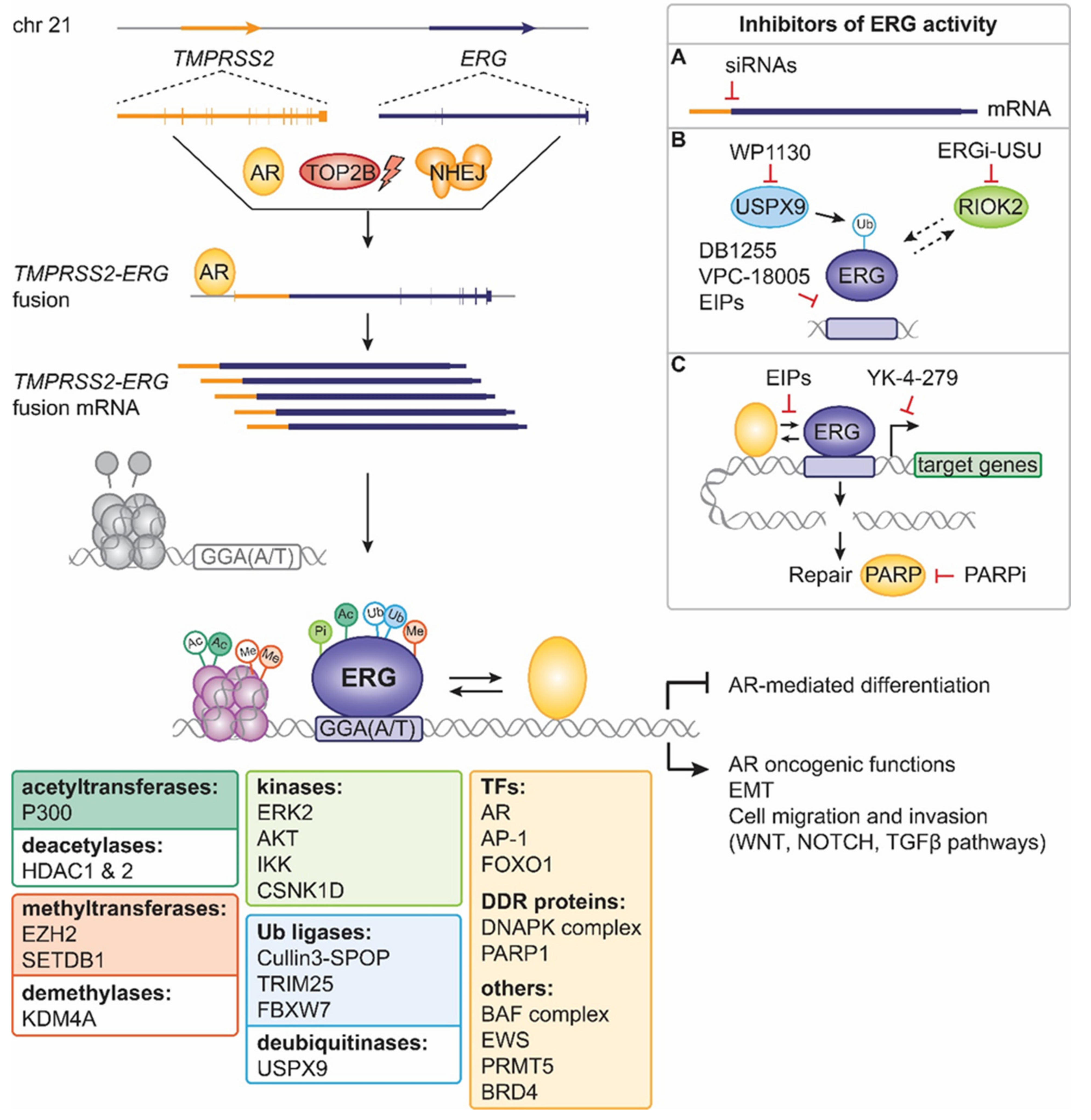
Cancers | Free Full-Text | Past, Current, and Future Strategies to Target ERG Fusion-Positive Prostate Cancer
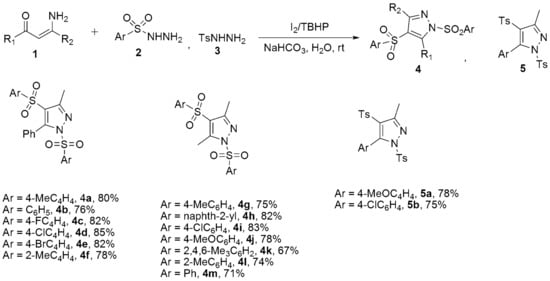
Biomedicines | Free Full-Text | A Review of the Recent Development in the Synthesis and Biological Evaluations of Pyrazole Derivatives

How does adenosine control neuronal dysfunction and neurodegeneration? - Cunha - 2016 - Journal of Neurochemistry - Wiley Online Library

Electrophilic Aminating Agents in Total Synthesis - O'Neil - 2021 - Angewandte Chemie International Edition - Wiley Online Library

Recent Developments in Photo‐Catalyzed/Promoted Synthesis of Indoles and Their Functionalization: Reactions and Mechanisms - Zheng - 2021 - Advanced Synthesis & Catalysis - Wiley Online Library

Biocatalytic Reduction Reactions from a Chemist's Perspective - Hollmann - 2021 - Angewandte Chemie International Edition - Wiley Online Library

Clip Chemistry: Diverse (Bio)(macro)molecular and Material Function through Breaking Covalent Bonds | Chemical Reviews