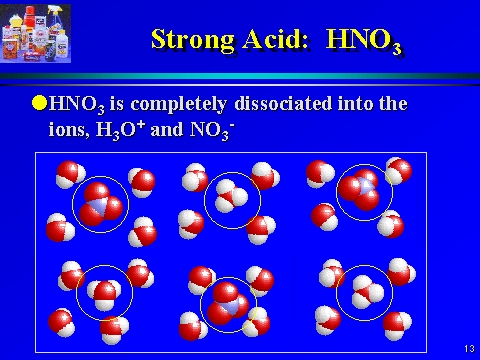
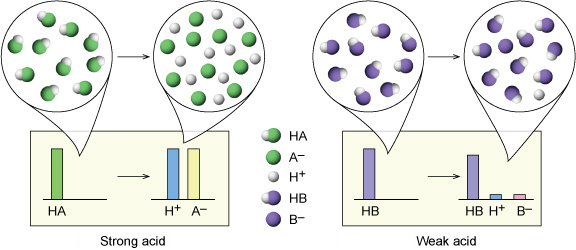
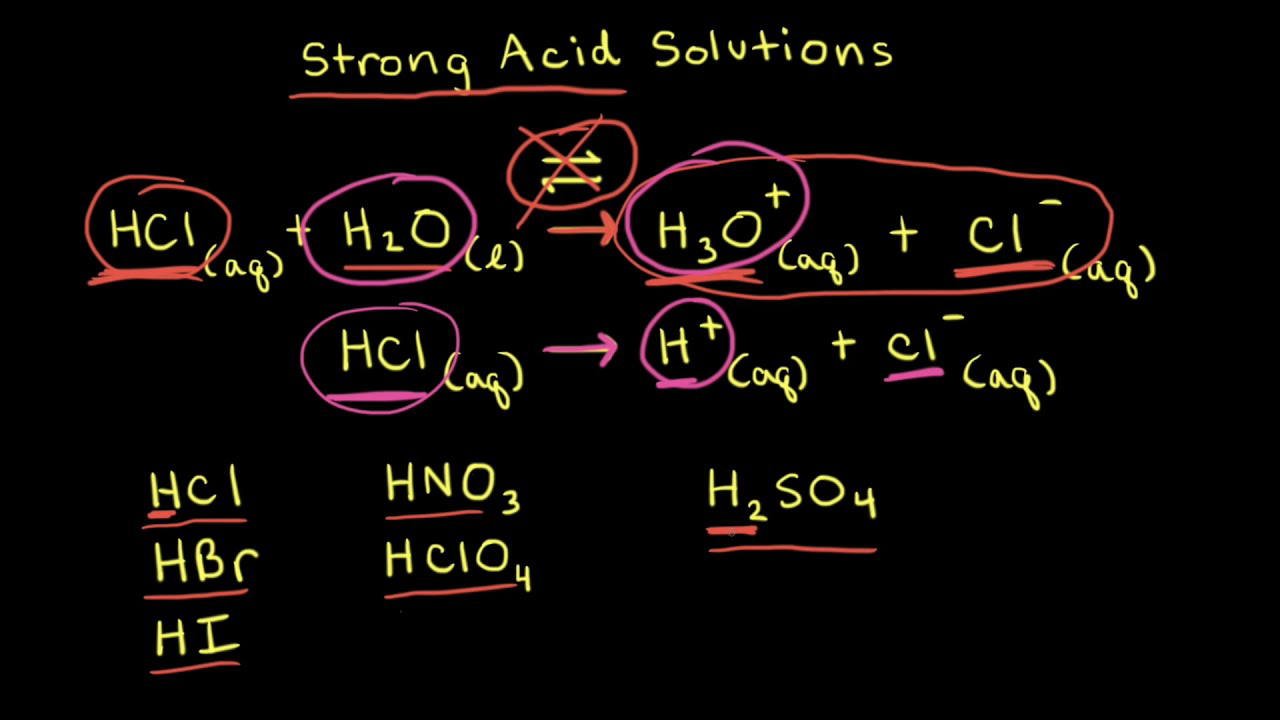
Why're hydrochloric acid, nitric acid, and sulfuric acid strong acids, while hydrofluoric acid and acetic acid weak acids?
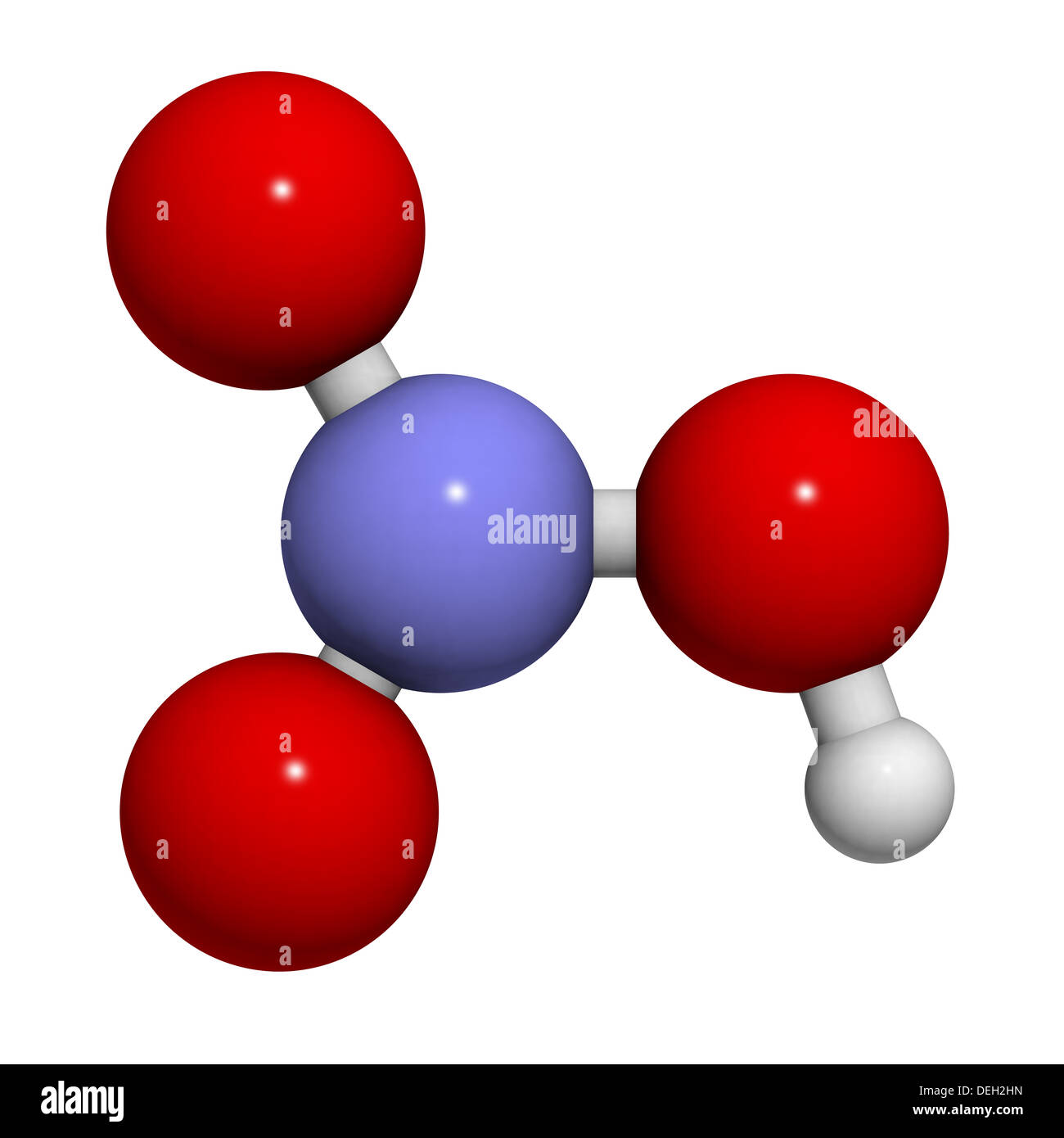
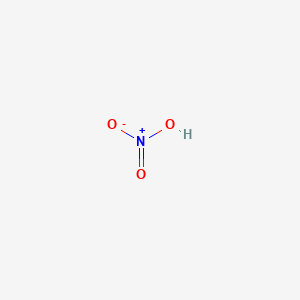
Nitric acid (HNO3) molecule, chemical structure. Strong and corrosive inorganic acid. Atoms are represented as spheres Stock Photo - Alamy
![SOLVED: Q: What are the [H+], [OH-], pH and pOH of a 0.002M solution of HNO3? Nitric acid v strong acid Completely ionizes in water HNO3 7 Ht + NO3 pH + SOLVED: Q: What are the [H+], [OH-], pH and pOH of a 0.002M solution of HNO3? Nitric acid v strong acid Completely ionizes in water HNO3 7 Ht + NO3 pH +](https://cdn.numerade.com/ask_images/096d2e96c5f04ffb9944004e8f1d5fba.jpg)
SOLVED: Q: What are the [H+], [OH-], pH and pOH of a 0.002M solution of HNO3? Nitric acid v strong acid Completely ionizes in water HNO3 7 Ht + NO3 pH +
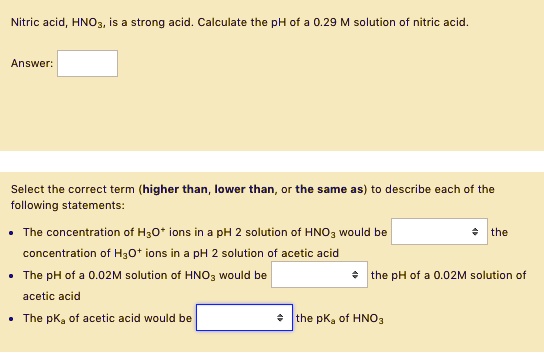
SOLVED: Nitric acid HNO3, is strong acid Calculate the pH of 0.29 M solution nitric acid Answer: Select the correct term (higher than, lower than following statements: the same as) describe each

Selective Separation of HNO3 and HCl by Extraction: The Investigation on the Noncovalent Interaction between Extractants and Acids by Density Functional Theory | The Journal of Physical Chemistry B
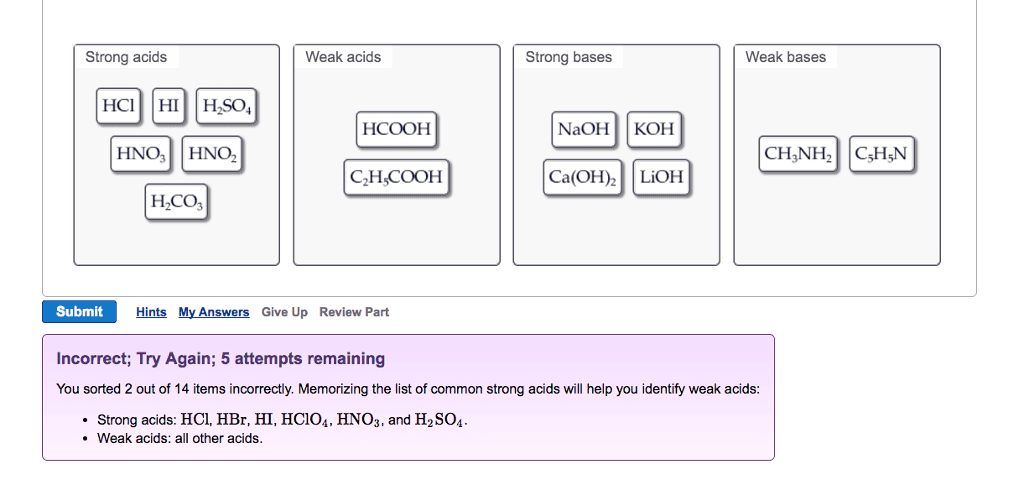
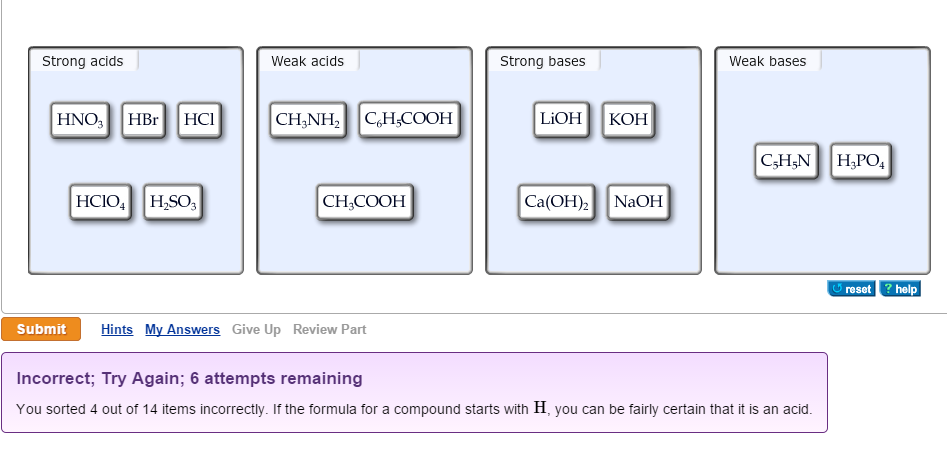
OneClass: Strong acids Weak acids Strong bases Weak bases NaOH KOH HCOOH HNO3 HNO2 CH3NH2 C5H5N C2H C...




:max_bytes(150000):strip_icc()/list-of-strong-and-weak-acids-603642-v2copy2-5b47abd0c9e77c001a395e55.png)