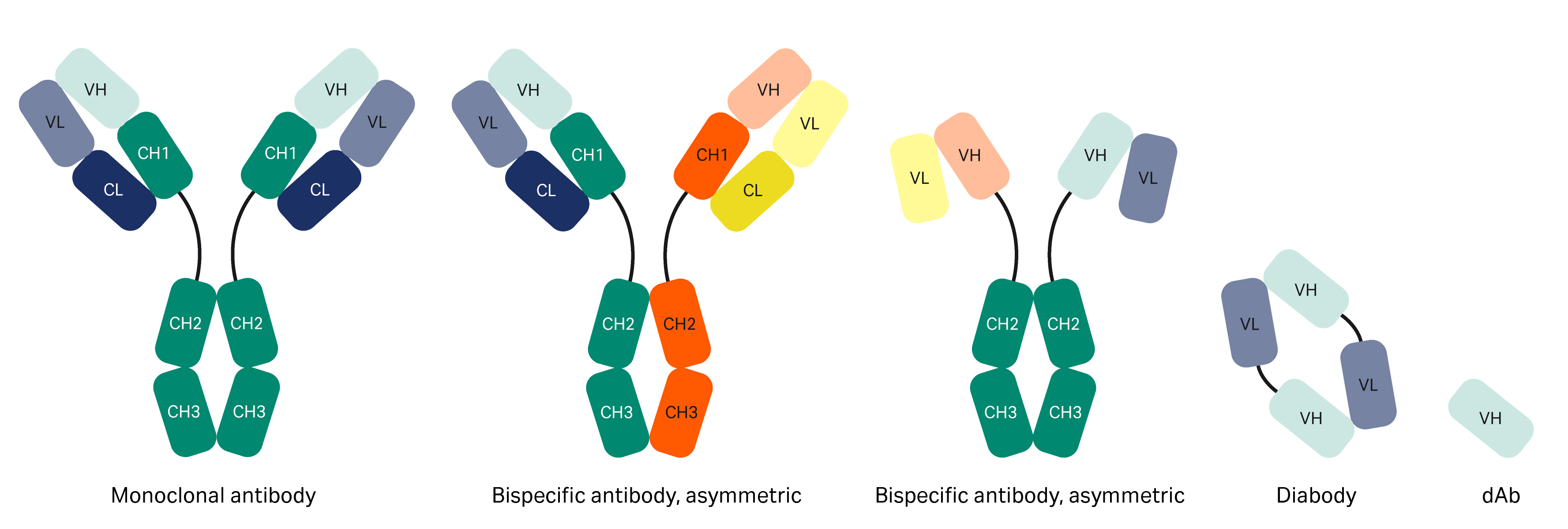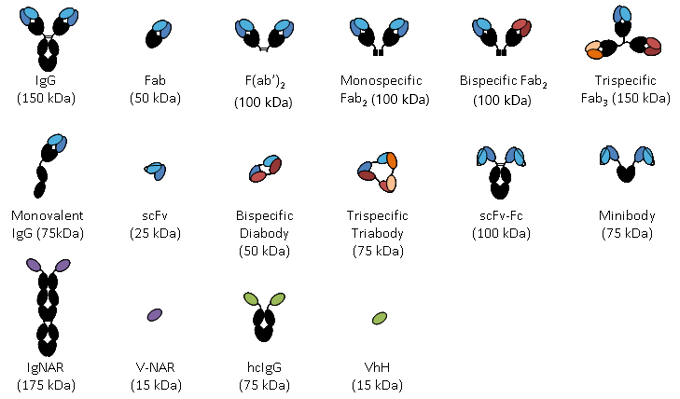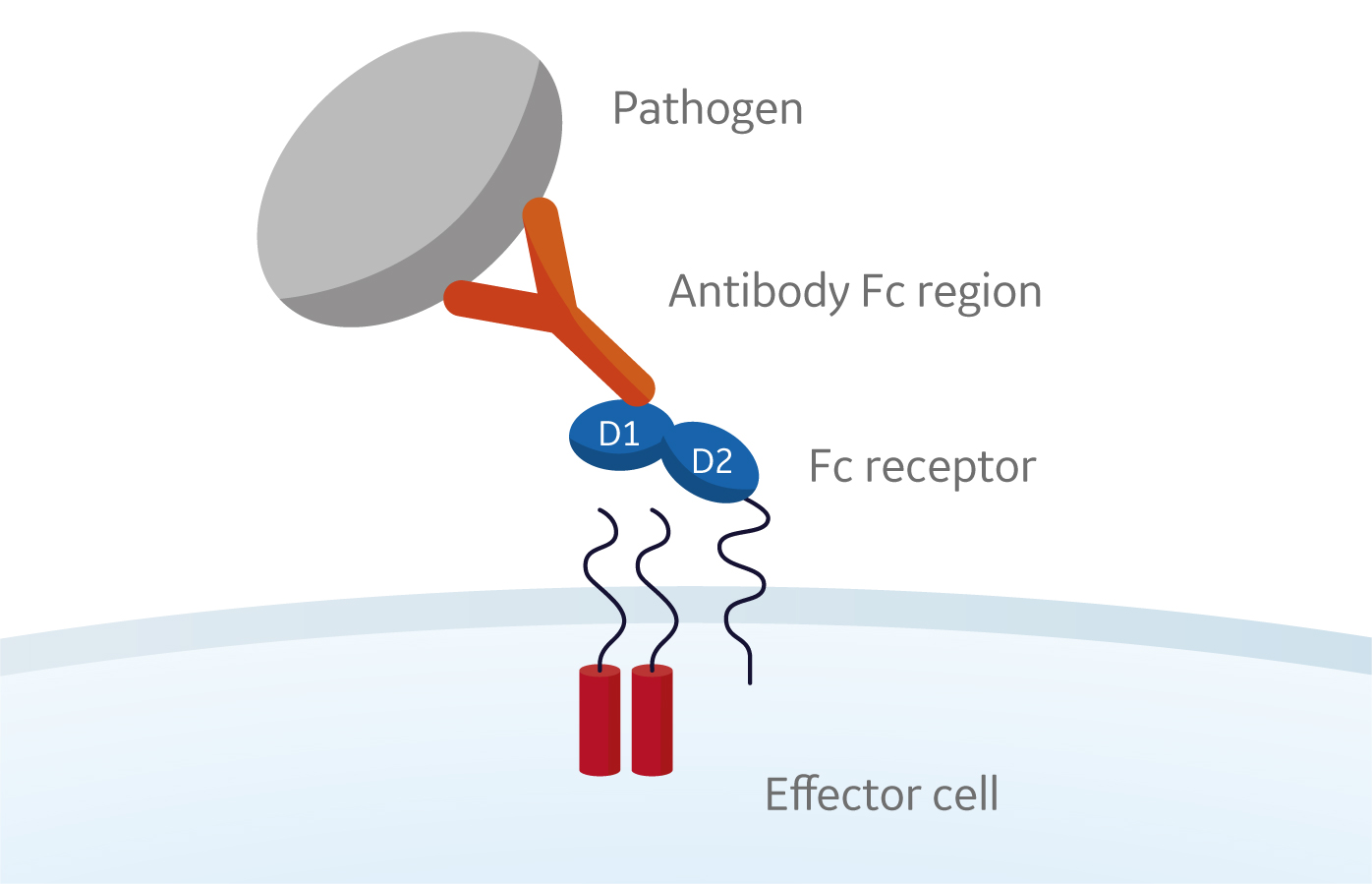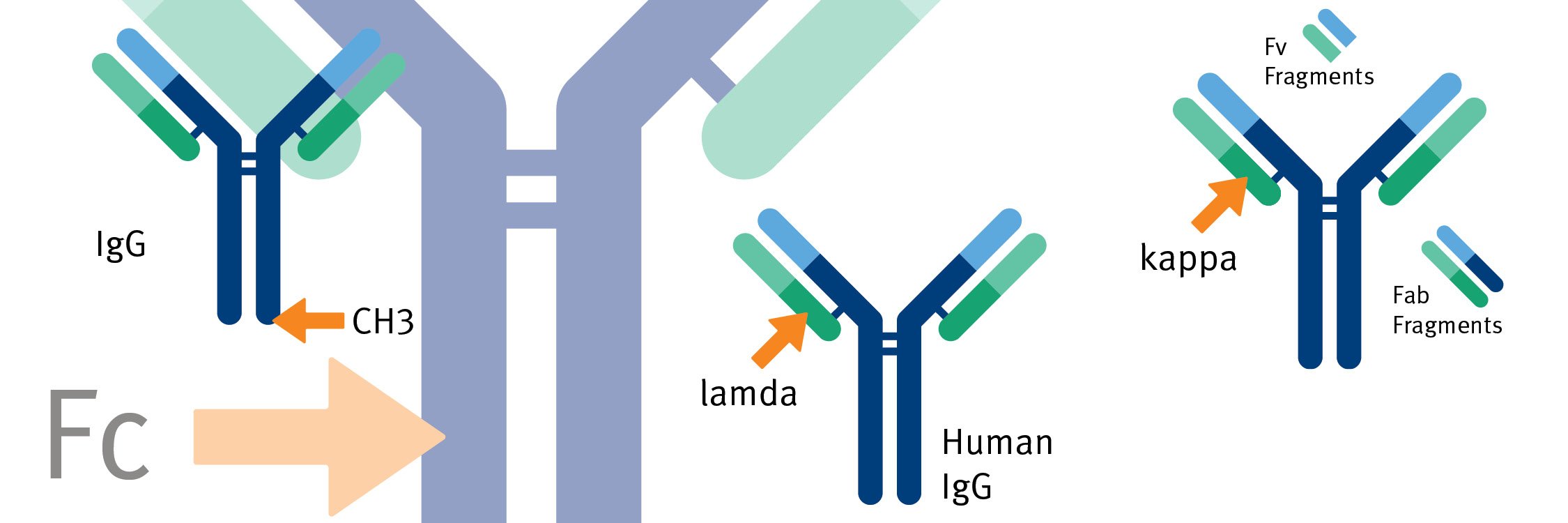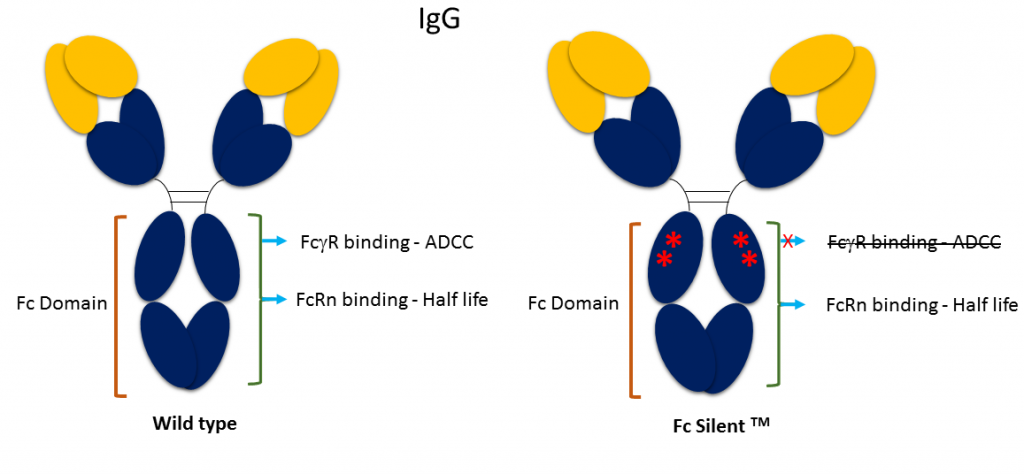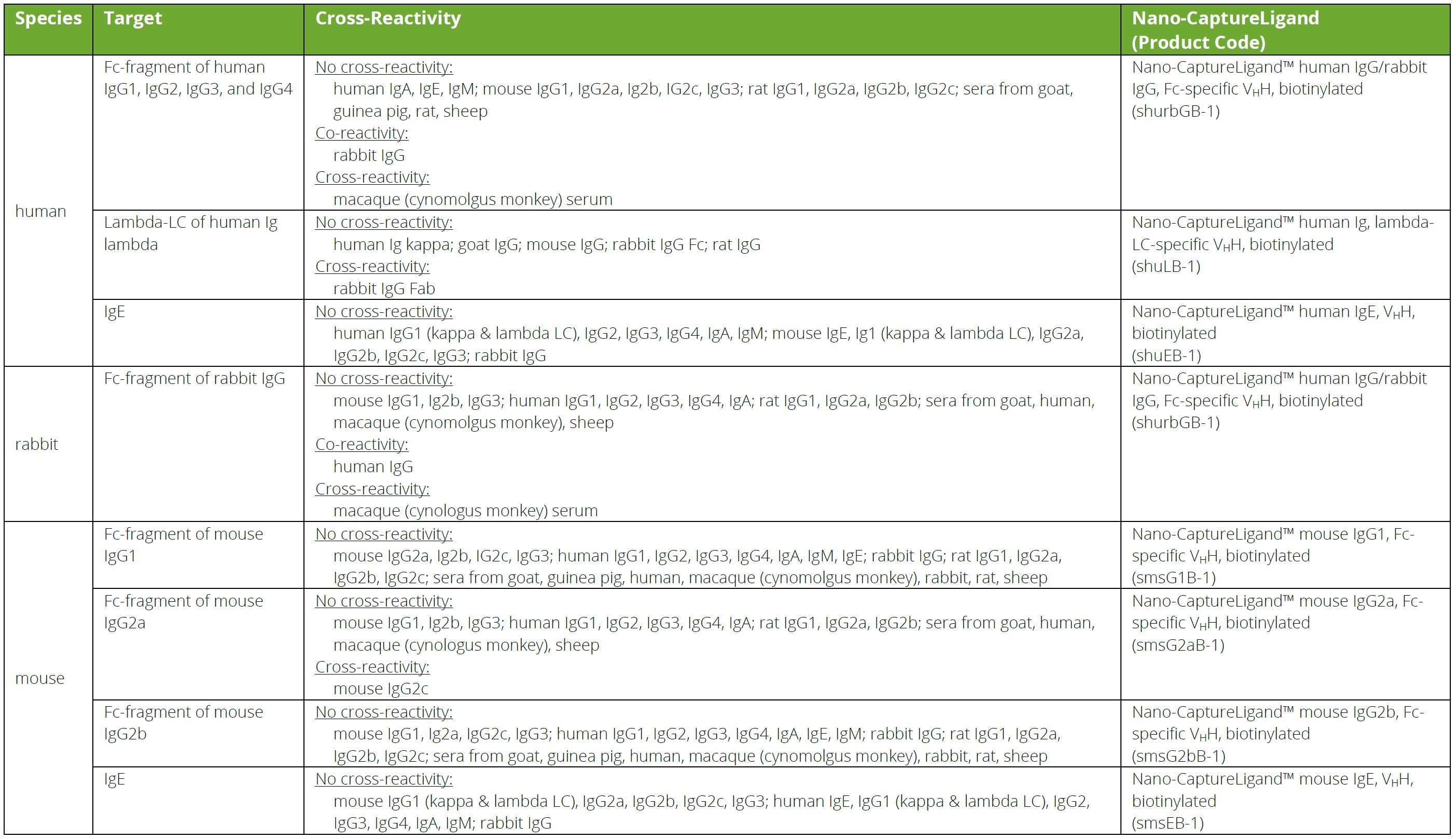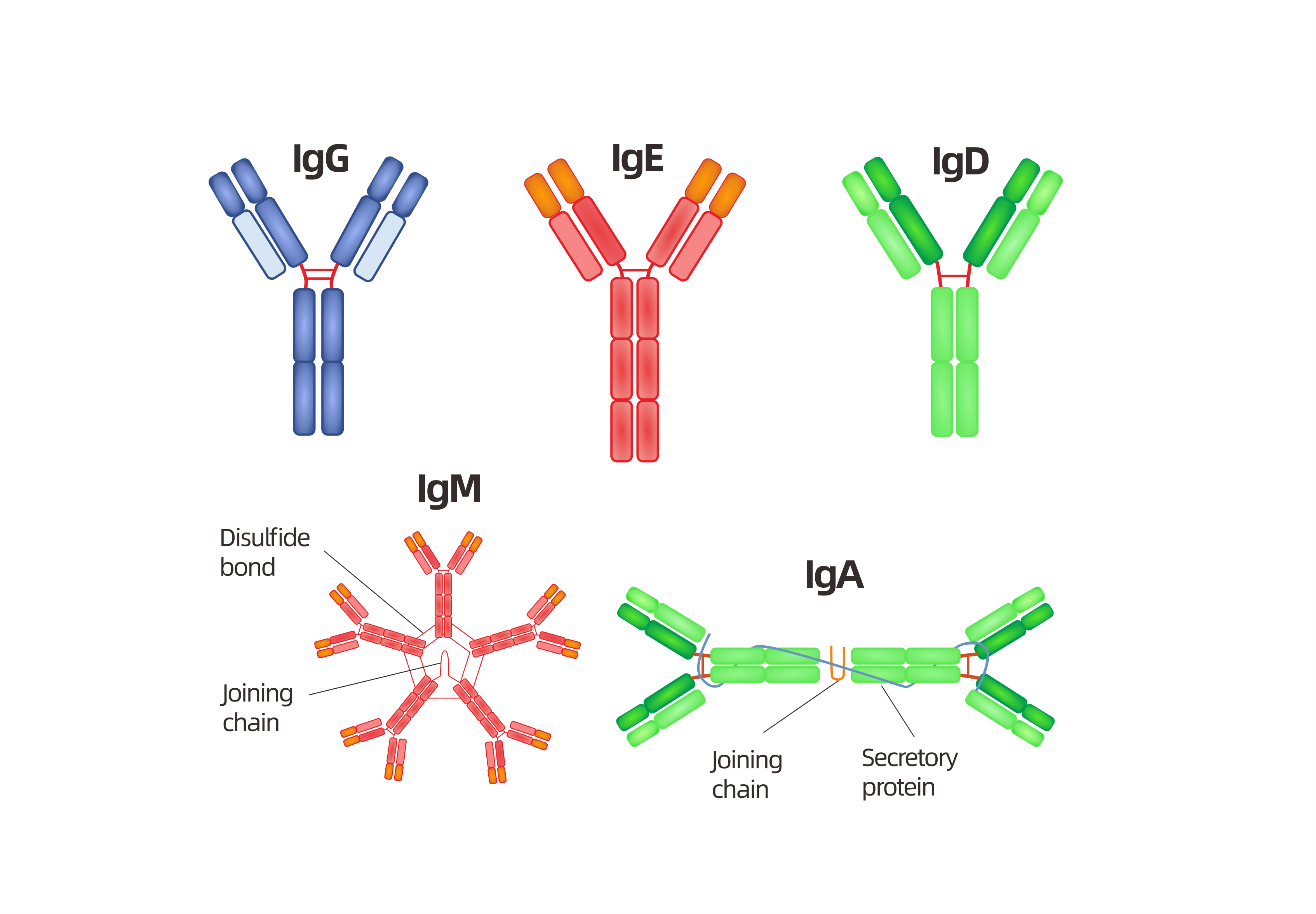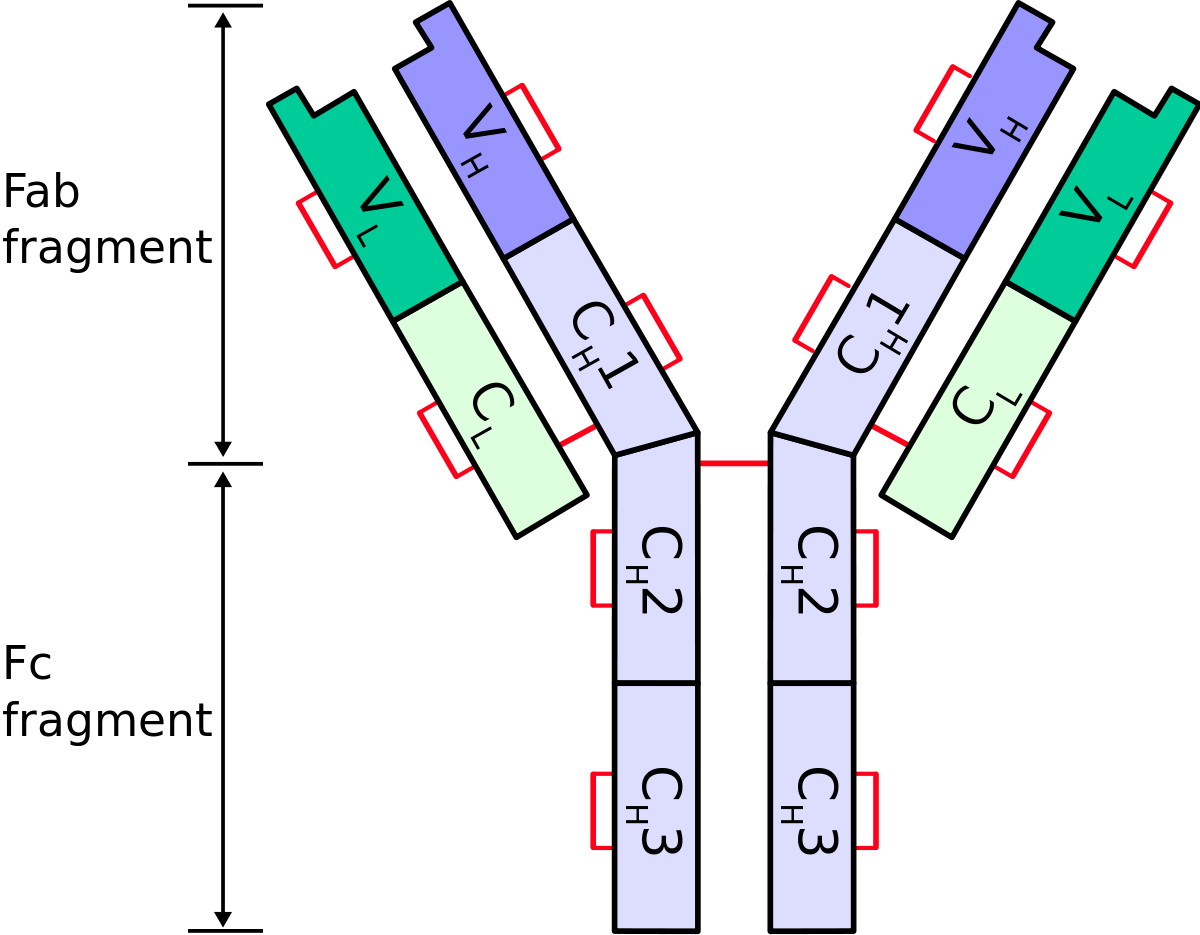
Frontiers | Engineering of Fc Fragments with Optimized Physicochemical Properties Implying Improvement of Clinical Potentials for Fc-Based Therapeutics

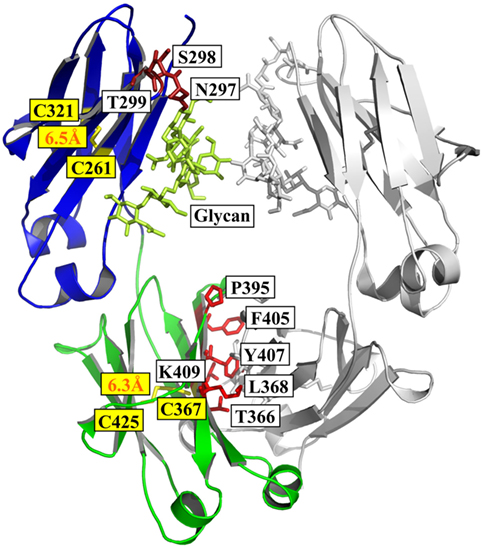
Schematic representation of homodimeric human IgG1-Fc (PDB-ID 1OQO),... | Download Scientific Diagram
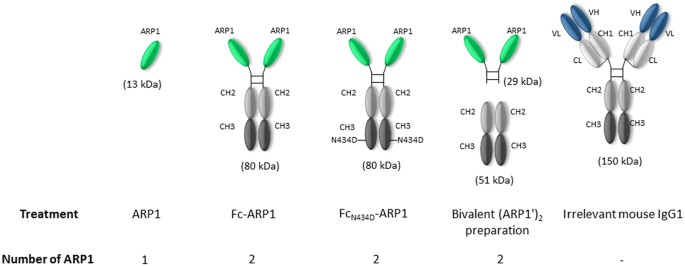
Fusion of the mouse IgG1 Fc domain to the VHH fragment (ARP1) enhances protection in a mouse model of rotavirus | Scientific Reports
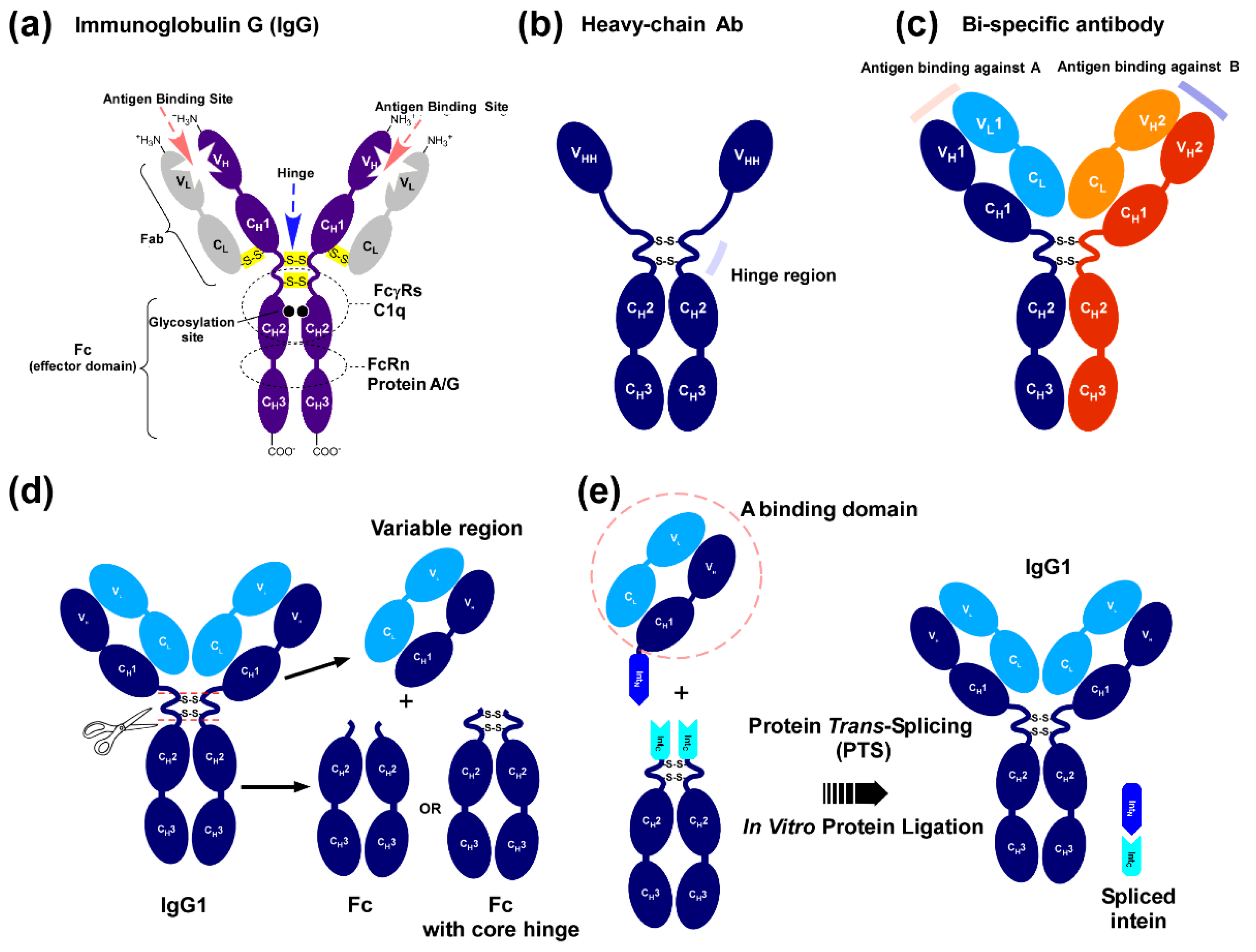
IJMS | Free Full-Text | An Off-the-Shelf Approach for the Production of Fc Fusion Proteins by Protein Trans-Splicing towards Generating a Lectibody In Vitro
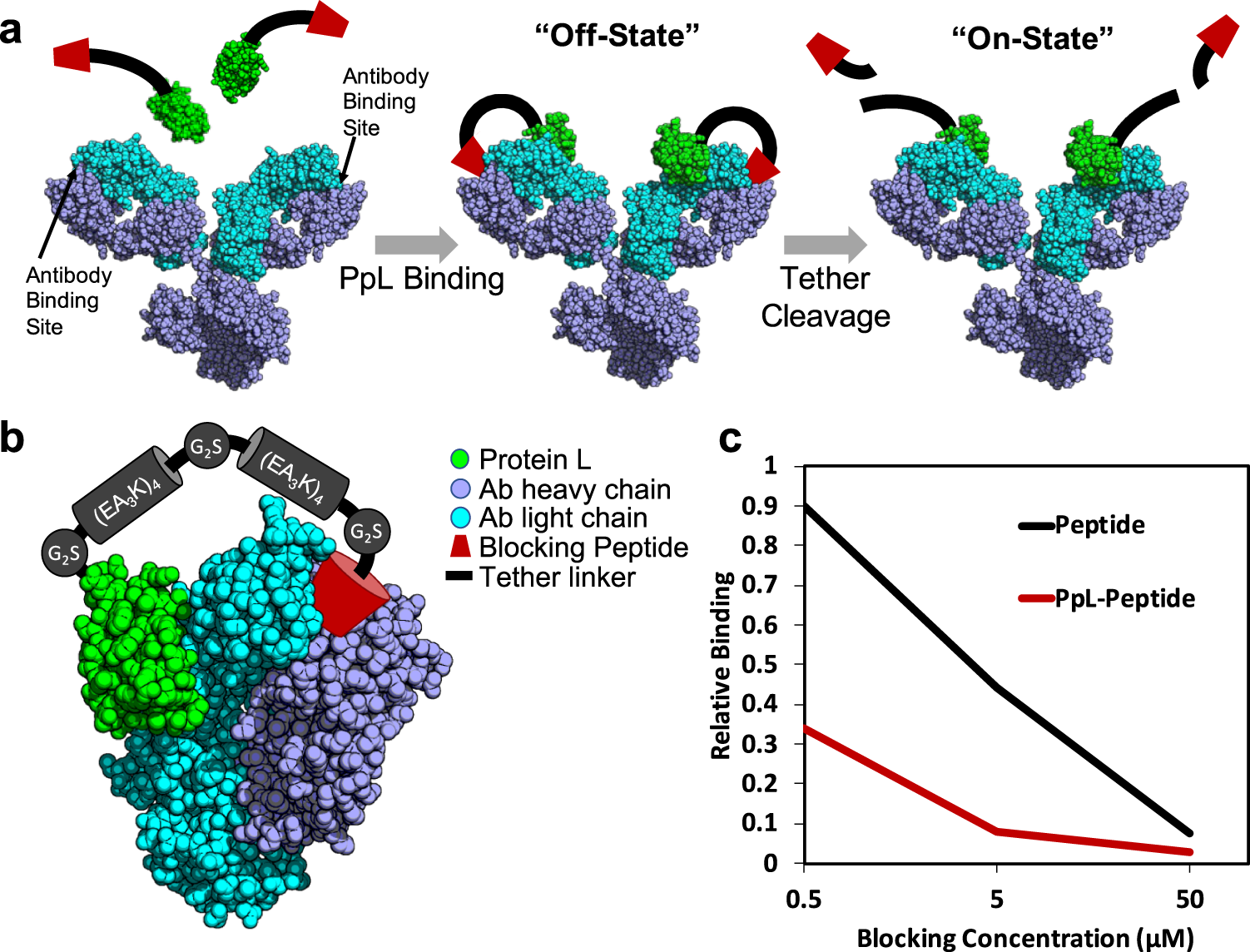
Turning antibodies off and on again using a covalently tethered blocking peptide | Communications Biology