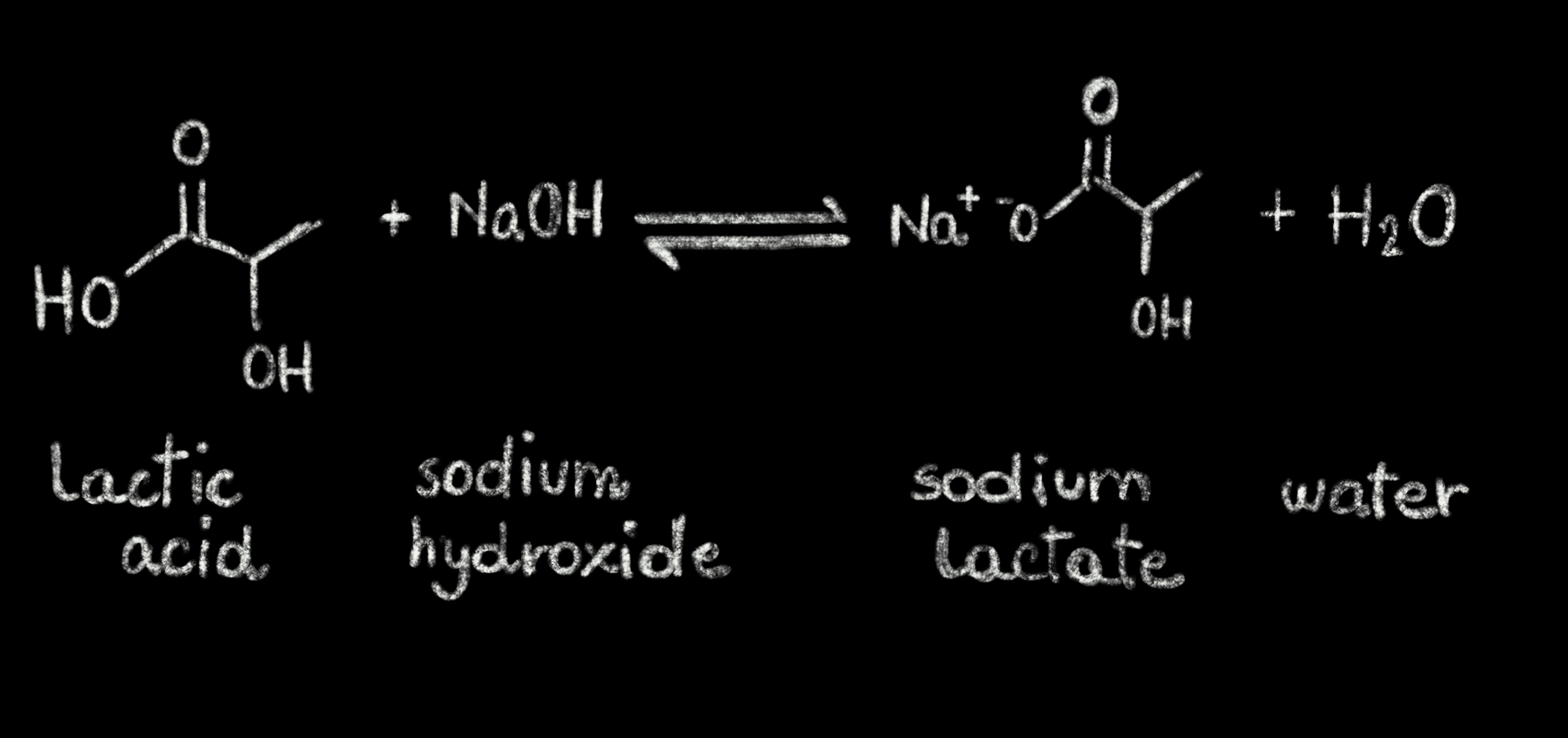
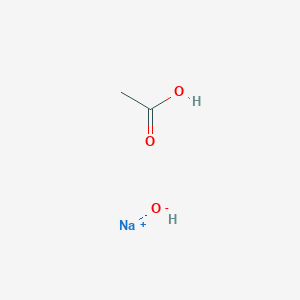
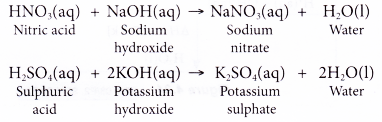Acids and Bases. Acids and bases Dissociation of water into hydrogen and hydroxide ions. pH is related to the concentration of hydrogen and hydroxide. - ppt download
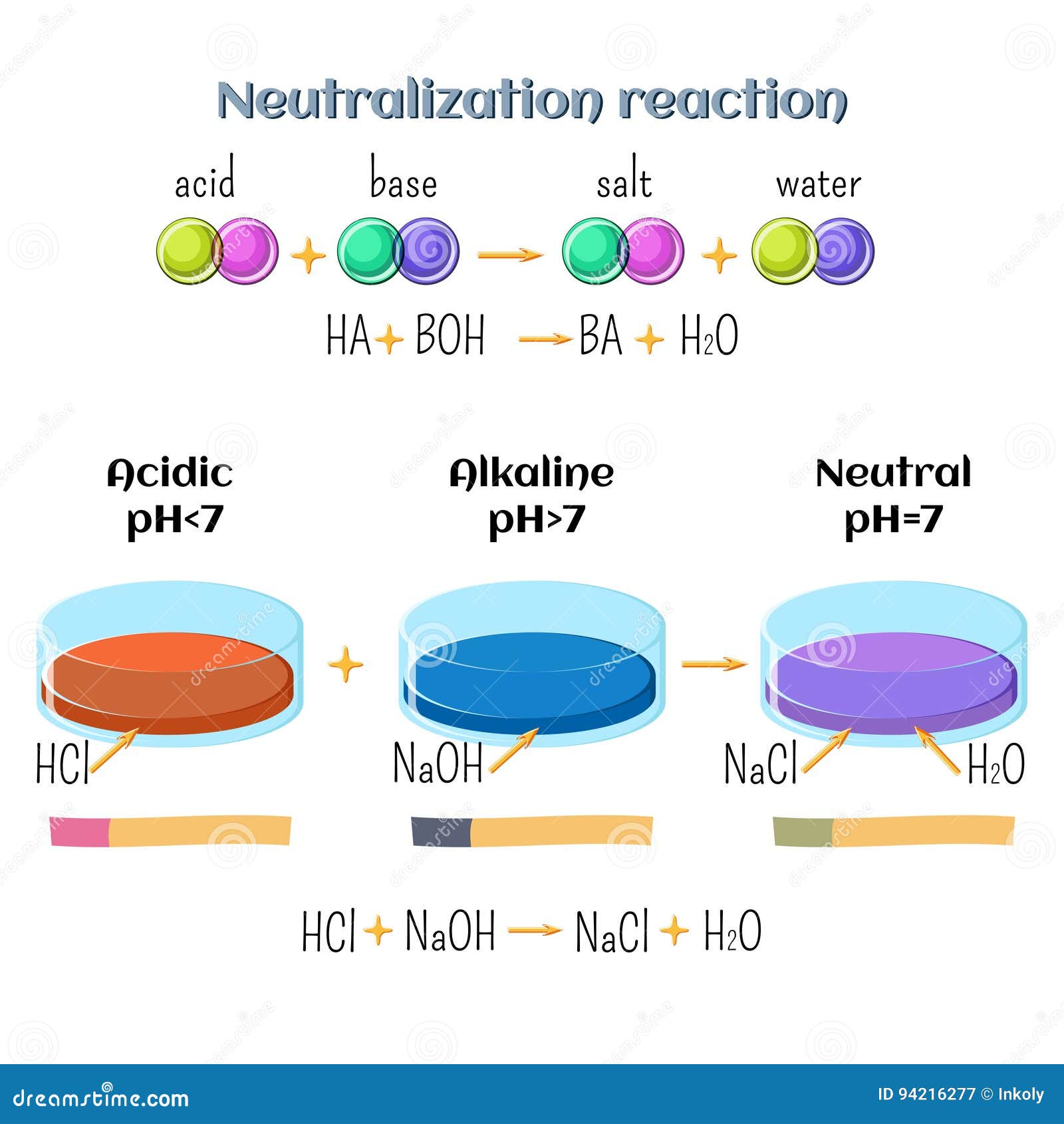
Acid-base, Neutralization Reaction of Hydrochloric Acid and Sodium Hydroxide. Types of Chemical Reactions, Part 6 of 7 Stock Vector - Illustration of acid, atom: 94216277

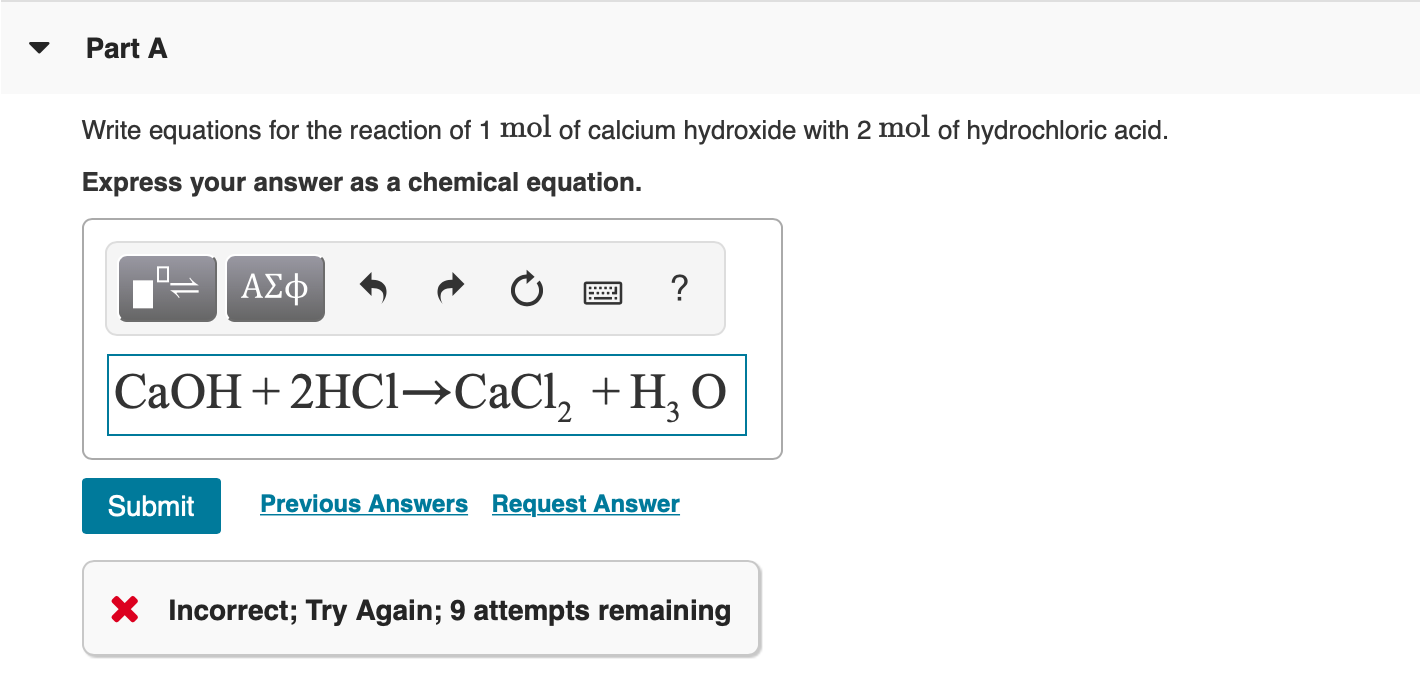
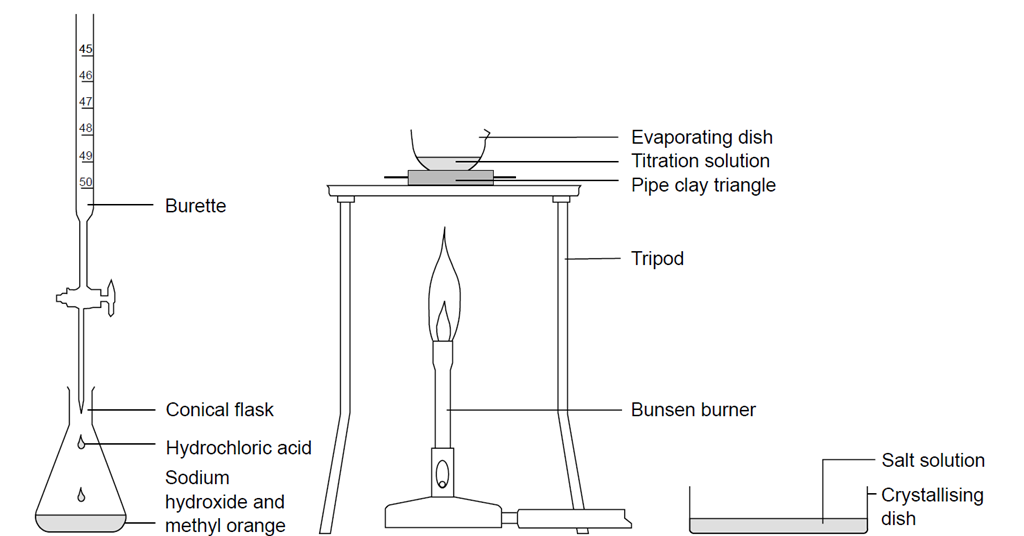
SOLVED: Question 20 (6 points) Tartaric acid, C4HsO reacts with sodium hydroxide according to the equation C4HsOs(aq) 2 NaOH(aq) NazCAH6O6(aq) HzO() If the sample of tartaric acld required 444.3 mL of 0.504

Acid-base, neutralization reaction of hydrochloric acid and sodium hydroxide. types of chemical reactions, part 6 of 7. | CanStock

Neutralizing Solutions with Sodium Hydroxide | Process & Chemical Formula - Video & Lesson Transcript | Study.com

Write a balanced chemical equation with state symbols for the reaction : sodium hydroxide solution(in water) reacts with hydrochloric acid solution(in water) to produce sodium chloride and water