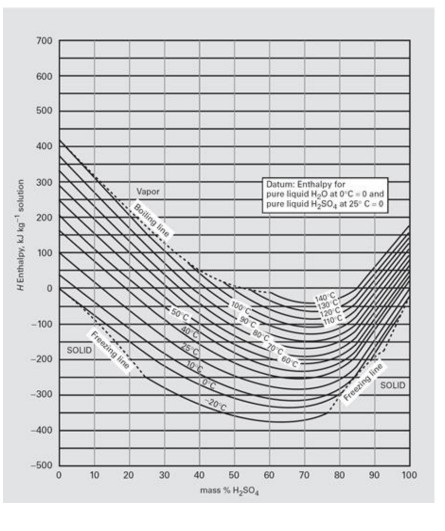
Physical properties and phase equilibrium of sulphuric acid (H2SO4) – Lauterbach Verfahrenstechnik GmbH
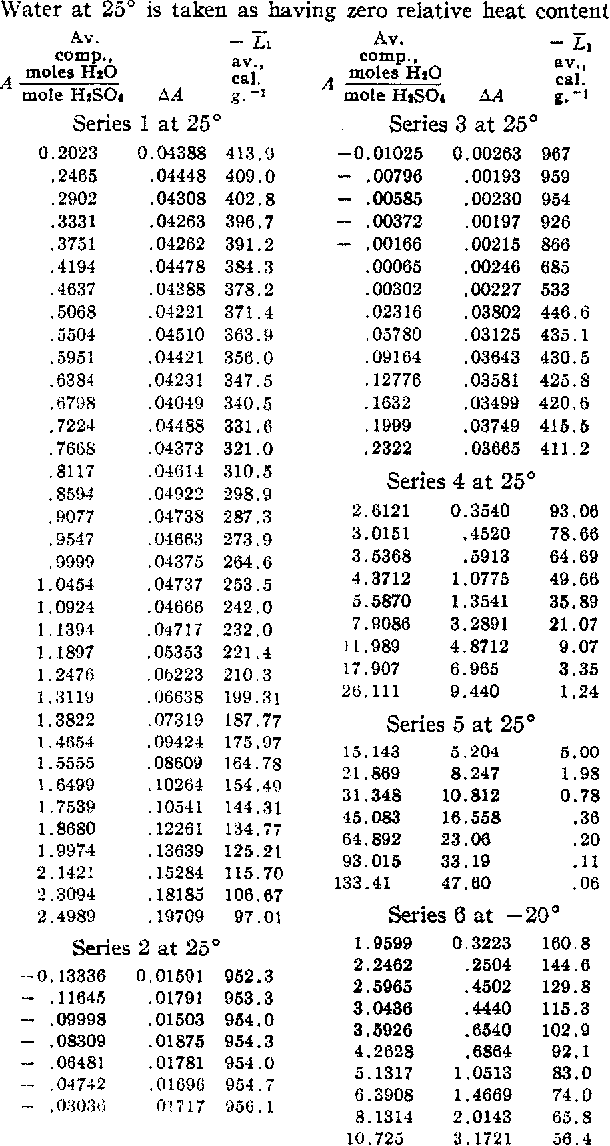
Table I from Aqueous Sulfuric Acid. Heat Capacity. Partial Specific Heat Content of Water at 25 and -20°1 | Semantic Scholar

SOLVED: A4.50 g sample of sulfuric acid (HSO4) is dissolved in 200.0 g water The temperature of solution rises from 22.00 %C to 33.89 %CCalculate the enthalpy of ionization rcactian of sulfuric

SOLVED: Be sure to answer all parts: Care must be taken when diluting sulfuric acid with water because the dilution process is highly exothermic: Hz0 HzSO4() HzSOA(aq) + heat (a Find AH

Table V from Aqueous Sulfuric Acid. Heat Capacity. Partial Specific Heat Content of Water at 25 and -20°1 | Semantic Scholar

Table I from Aqueous Sulfuric Acid. Heat Capacity. Partial Specific Heat Content of Water at 25 and -20°1 | Semantic Scholar

Figure 2 from Aqueous Sulfuric Acid. Heat Capacity. Partial Specific Heat Content of Water at 25 and -20°1 | Semantic Scholar

SOLVED: Diluting sulfuric acid with water is highly exothermic: H,SOA() H! HSOs(aq) heat (Use data from the Appendix to find AHG for diluting 1.00 mol of HzSO4() (d 1.83 g/mL) to 1




![3] The heat of H2SO4 dissolving in the water. | Download Table 3] The heat of H2SO4 dissolving in the water. | Download Table](https://www.researchgate.net/publication/276272665/figure/tbl1/AS:669453972959252@1536621673863/3-The-heat-of-H2SO4-dissolving-in-the-water.png)