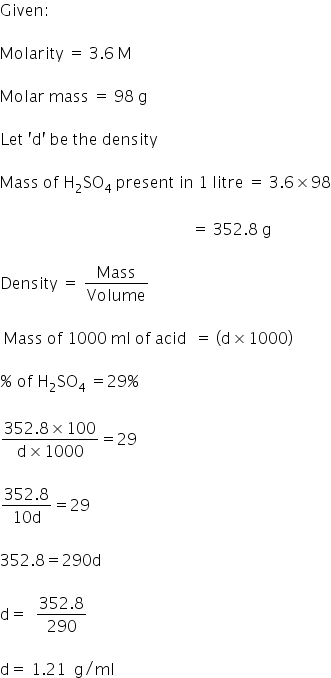Kinetics of selenium and tellurium removal with cuprous ion from copper sulfate-sulfuric acid solution | Semantic Scholar

b7.docx - 1. The density of concentrated sulphuric acid is 1.8 g/cm3. Calculate the volume of 3.1 kg of the acid. Solution Volume = mass / density = | Course Hero
What is the volume of concentrated H2SO4 of specific gravity 1.84 and containing 98% H2SO4 by weights that would contain 40 gm of pure H2SO4? - Quora
The density (in g mL^–1 ) of a 3.60M sulphuric acid solution that is 29% H2SO4 (molar mass = 98g mol^–1 ) by mass will be. - Sarthaks eConnect | Largest Online Education Community

a bottle of concentrated sulphuric acid (density of 1.80 g cm-3) is labelled as 86% as weight. What is - Brainly.in
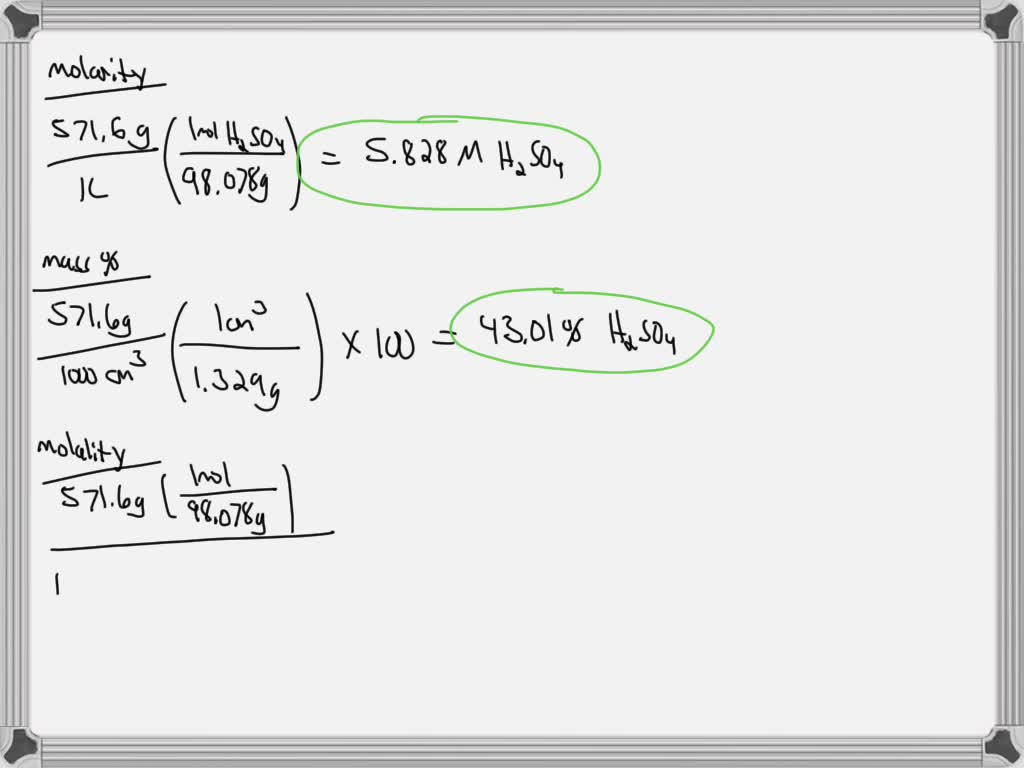
SOLVED: Molarity of sulphuric acid is 0.8 and its density is 1.06 gram per centimetre cube What will be its concentration in terms of molarity and mole fraction? A. 0.785 mole per

The specific gravity of sulphuric acid 1.8. What volume of this sample of concentrated H 2 SO 4 is required to prepare 500 ml 0.9 M H 2 SO 4 solution?
reactivity - How could mass increase when sulfuric acid is added to calcium carbonate? - Chemistry Stack Exchange
![The density (in g ml^-1 ) of a 3.60 M sulphuric acid solution having 29% H2SO4 [molar mass = 98 g mol^-1 ] by mass, will be: The density (in g ml^-1 ) of a 3.60 M sulphuric acid solution having 29% H2SO4 [molar mass = 98 g mol^-1 ] by mass, will be:](https://dwes9vv9u0550.cloudfront.net/images/5468271/2ef79989-6c93-4eae-b4f4-8ef6608caa27.jpg)