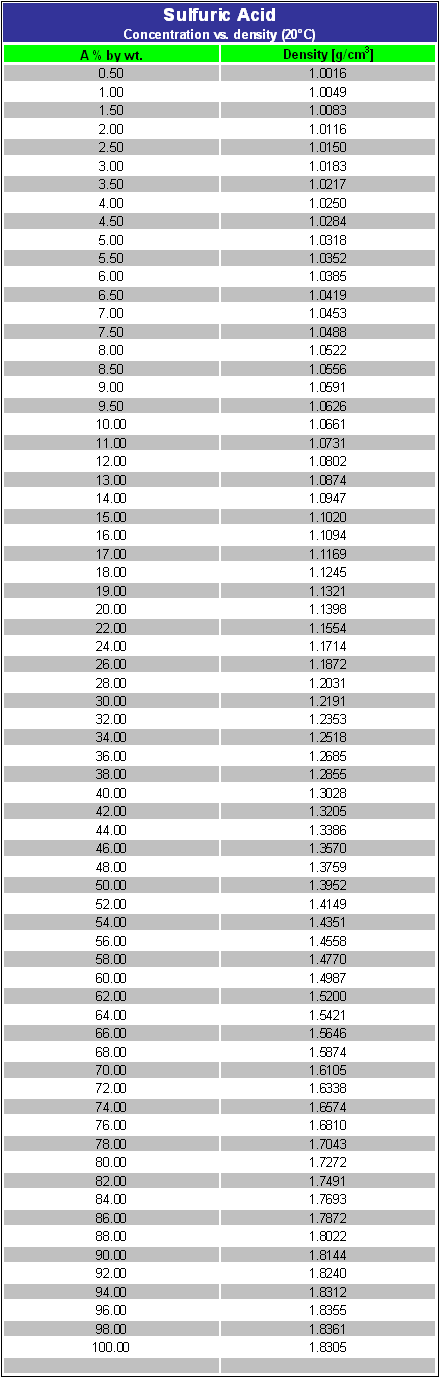
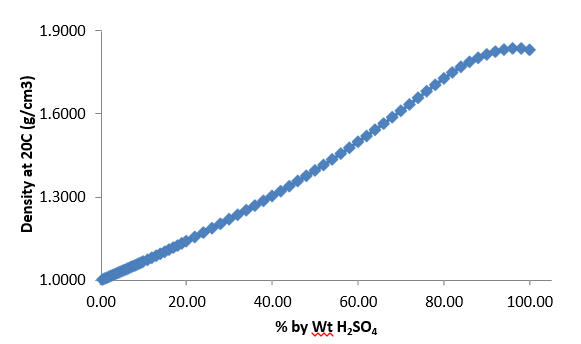
What is the molarity of concentrated sulfuric acid if it is 96% by mass H2so4 and has a density of 1.84g/mL? - Quora

Laboratory grade concentrated sulphuric acid has a density 1.82g cc^(-1) .Weight percentage of acid is 98. Calculate the normality of solution.

Concentrated H2SO4 has a density 1.9g/ml and is 99% H2SO4 by mass. Calculate the molarity. - YouTube

SOLVED:A chemist needs 35.0 g of concentrated sulfuric acid for an experiment. The density of concentrated sulfuric acid at room temperature is 1.84 g / mL. What volume of the acid is required?

Concentrated sulphuric acid has density of 1.9 g/mL and 99% H2SO4 by mass. Calculate the molarity of the acid.

Ma'am, could you please solve this question- Concentrated sulphuric acid is 98% H2SO4 by mass and has a density of - Chemistry - - 14853829 | Meritnation.com
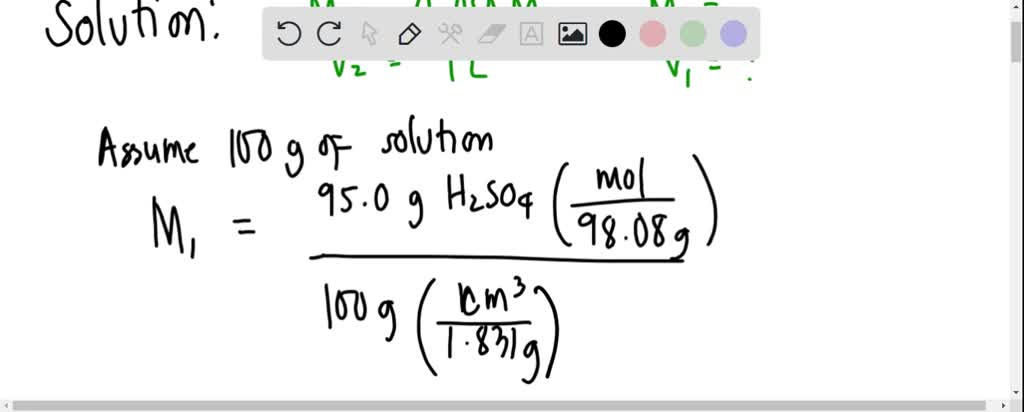
SOLVED: Calculate the volume (in millilitres) of concentrated sulfuric acid, 95.0% (g/100 solution) with a density of 1.831 g/cm3 required to prepare 1 L of a 0.050 M solution.