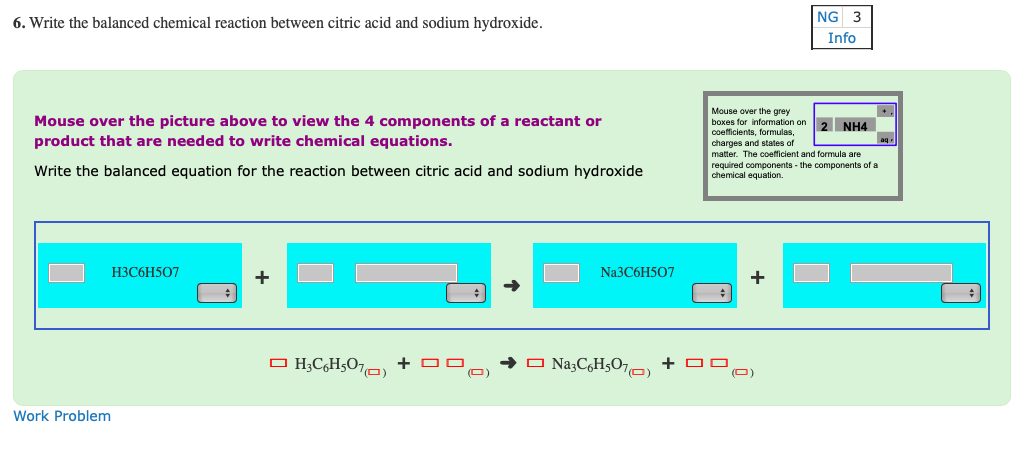
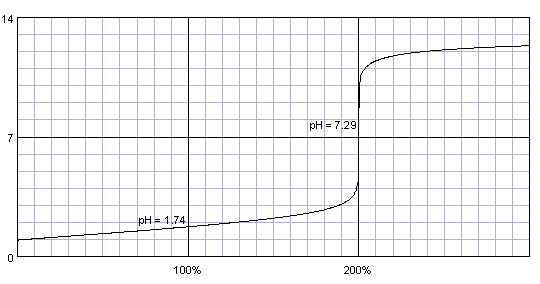
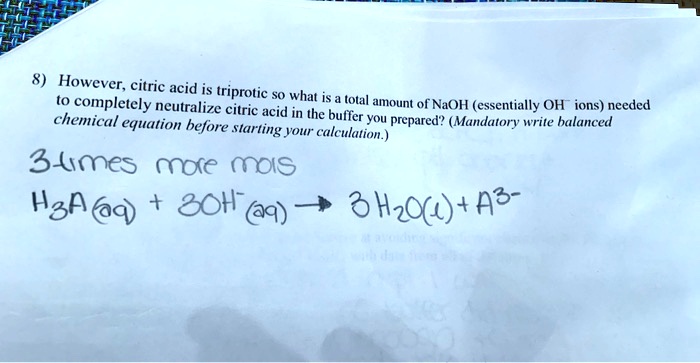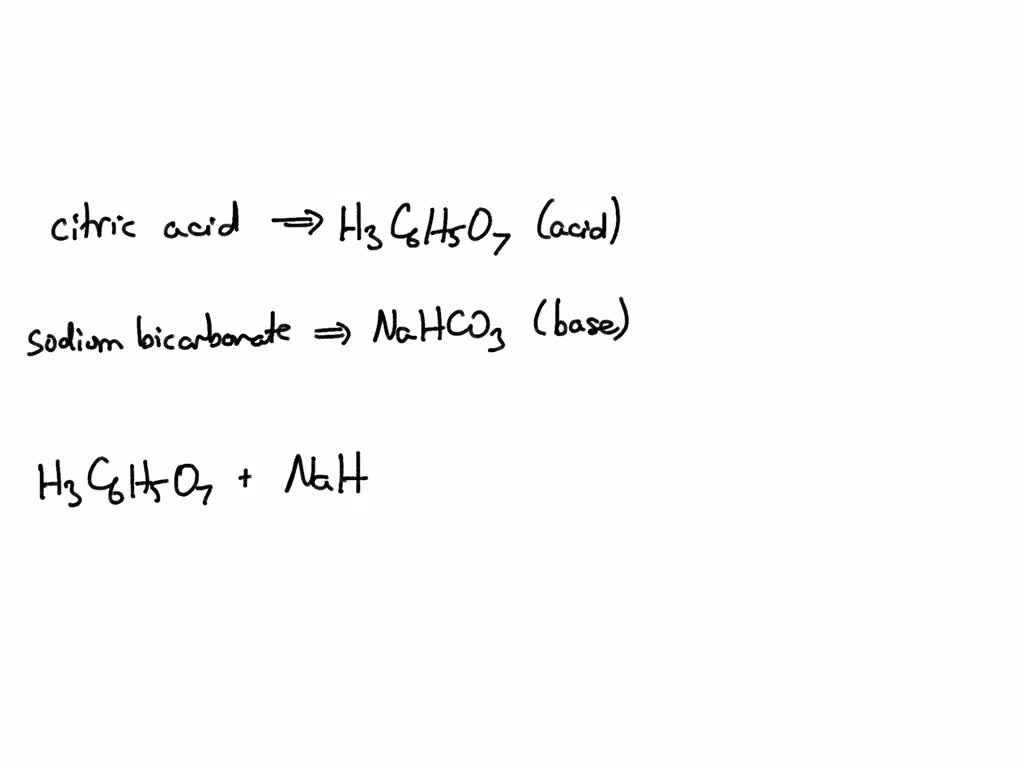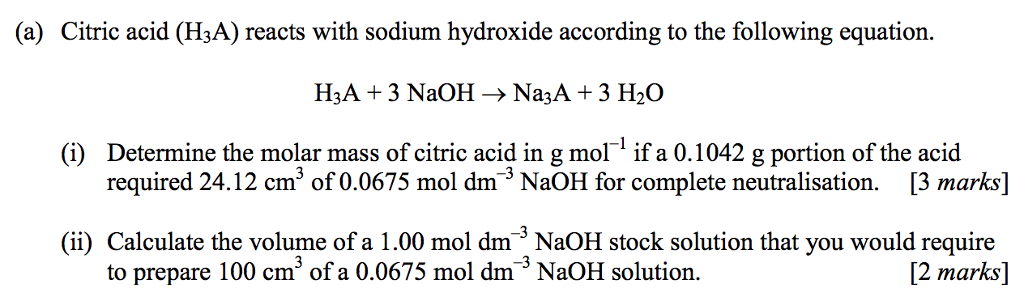Write out a detailed procedure for determining the enthalpy of reaction between citric acid (H3Cit) and sodium hydroxide (NaOH). Your will be given 0.50M solutions of citric acid and sodium hydroxide.
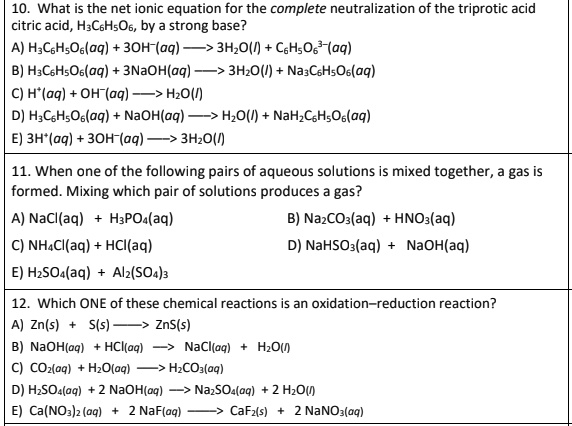
SOLVED: What is the net ionic equation for the complete neutralization of the triprotic acid citric acid, H-CbHsOb, by strong base? A) HyCsH;Os(aq) 30H-(aq) 3HzO() CsHsOs-(aq) B) H-CsHsOs(aq) 3NaOH(aq) 5s 3HzO() -

OneClass: Write the balanced net ionic equation for the reaction between citric acid and sodium hydro...

Buffer Solution (citric acid, sodium hydroxide, hydrogen chloride) tracable to SRM from NIST and PTB pH 2.00 (25°C) Certipur® | Sigma-Aldrich