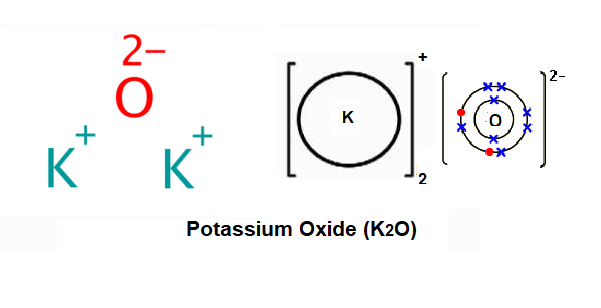
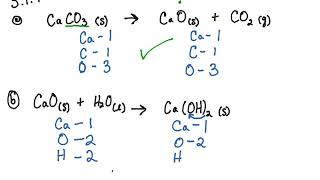SOLVED: Ammonia gas can be prepared by the reaction of a metal oxide such as calcium oxide with ammonium chloride: CaO(s) + 2 NH4 Cl(s) 72 NH (g) + HzO(g) + Ca

SOLVED: Caldum oxide reacts with water in combination reaction produce calcium hydroxide; CaO (s) H2O (I) Ca(OH)2 (s) 3.50-8 sample of CaO is reacted with 3.38 gof HzO How Many grams of

Question Video: Calculating the Mass of Calcium Carbonate Required to Produce a Given Mass of Calcium Oxide | Nagwa

Calculate the lattice enthalpy of calcium oxide from the following data. Data found in the IB chemistry data booklet and the Born Haber cycle drawn in part (a) enthalpy of atomisation of