Topical bioequivalence: Experimental and regulatory considerations following formulation complexity - ScienceDirect

Percent of studies passing bioequivalence (BE) (power curves); average... | Download Scientific Diagram

PDF) Statistical Design Based on 90 % Confidence Intervals Analysis of Bioequivalence Studies of Sustained Release Capsules of Metoprolol Tartrate

PDF) Pooled bioequivalence study database from Turkey: characterization of adverse events and determination of split points based on Gini Index as a promising method

A Randomized, Crossover, Single-Dose Bioequivalence Study of Two Extended Release Tablets of Donepezil 23 mg in Healthy Human Volunteers under Fasting and Fed State – topic of research paper in Clinical medicine.

PDF) Between-Batch Pharmacokinetic Variability Inflates Type I Error Rate in Conventional Bioequivalence Trials: A Randomized Advair Diskus® Clinical Trial
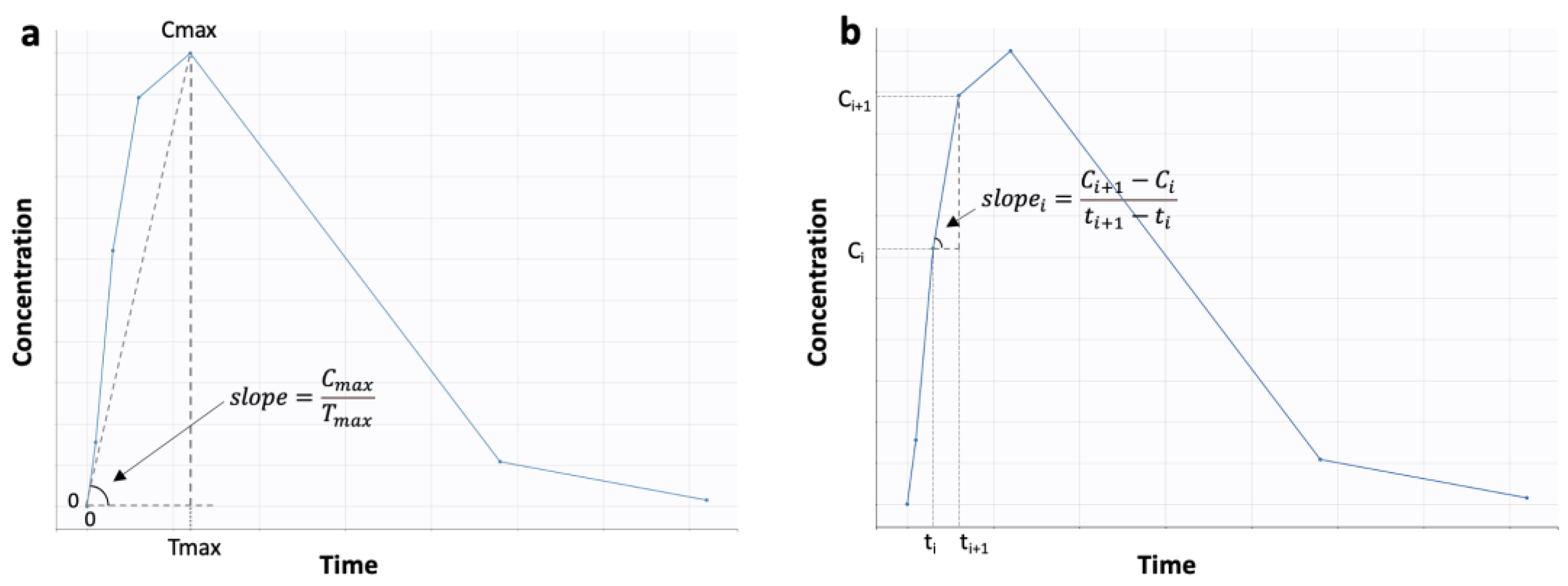
Applied Sciences | Free Full-Text | On the Interplay between Machine Learning, Population Pharmacokinetics, and Bioequivalence to Introduce Average Slope as a New Measure for Absorption Rate
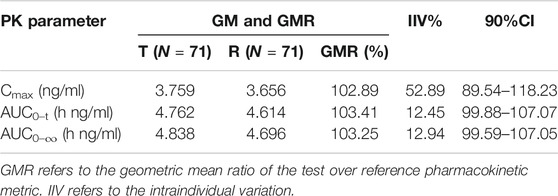
Frontiers | Pharmacokinetics and Bioequivalence of Rasagiline Tablets in Chinese Healthy Subjects Under Fasting and Fed Conditions: An Open, Randomized, Single-Dose, Double-Cycle, Two-Sequence, Crossover Trial
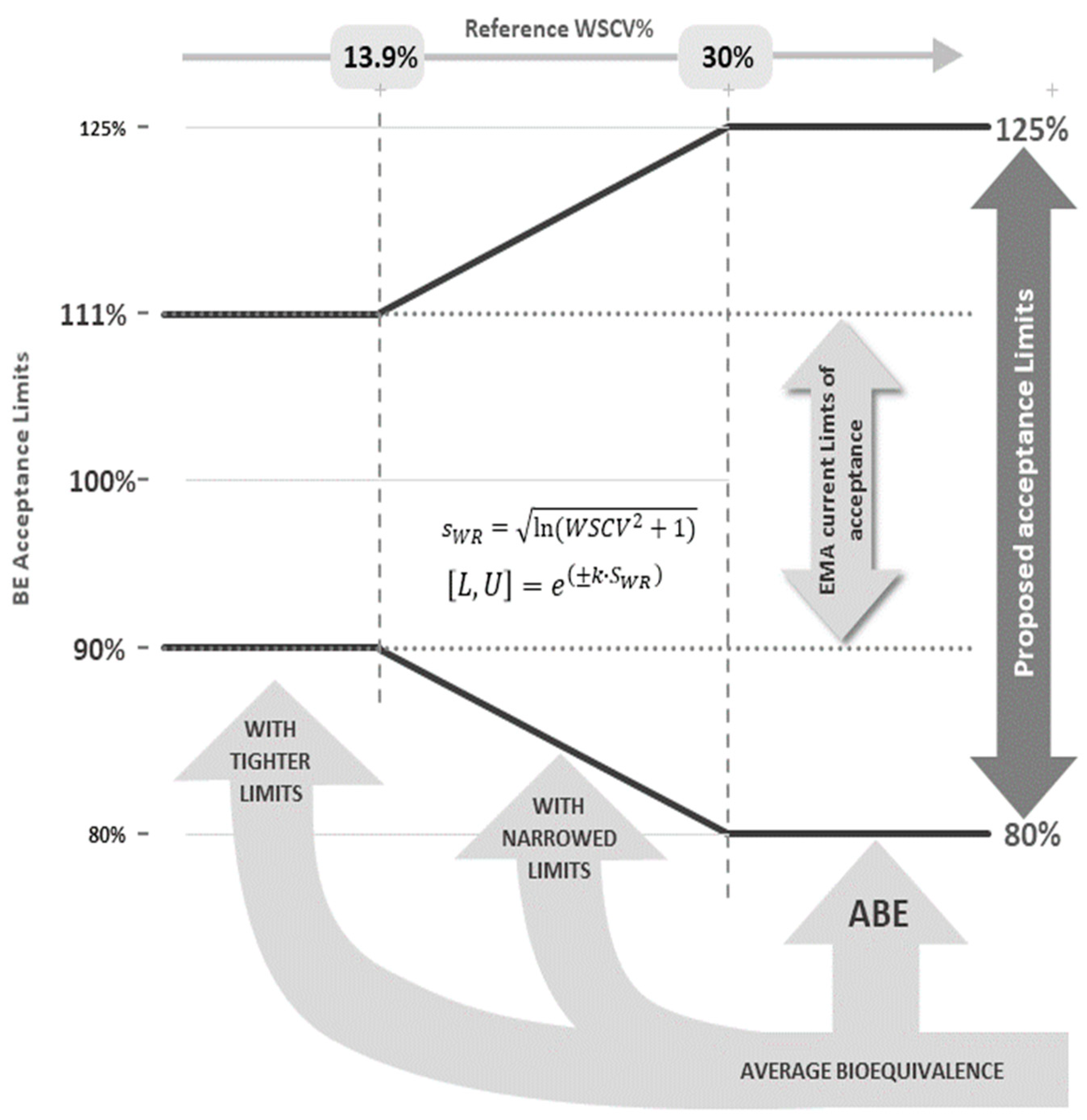
Pharmaceutics | Free Full-Text | Evaluation of a Proposed Approach for the Determination of the Bioequivalence Acceptance Range for Narrow Therapeutic Index Drugs in the European Union

The 80–125% BE limits are represented along the x-axis as two " goal... | Download Scientific Diagram

Between-Batch Bioequivalence (BBE): a Statistical Test to Evaluate In Vitro Bioequivalence Considering the Between-Batch Variability | SpringerLink

In Vitro Predictive Dissolution Test Should Be Developed and Recommended as a Bioequivalence Standard for the Immediate-Release Solid Oral Dosage Forms of the Highly Variable Mycophenolate Mofetil | Molecular Pharmaceutics

Adjusted Indirect Treatment Comparison of the Bioavailability of WHO‐Prequalified First‐Line Generic Antituberculosis Medicines - Gwaza - 2014 - Clinical Pharmacology & Therapeutics - Wiley Online Library
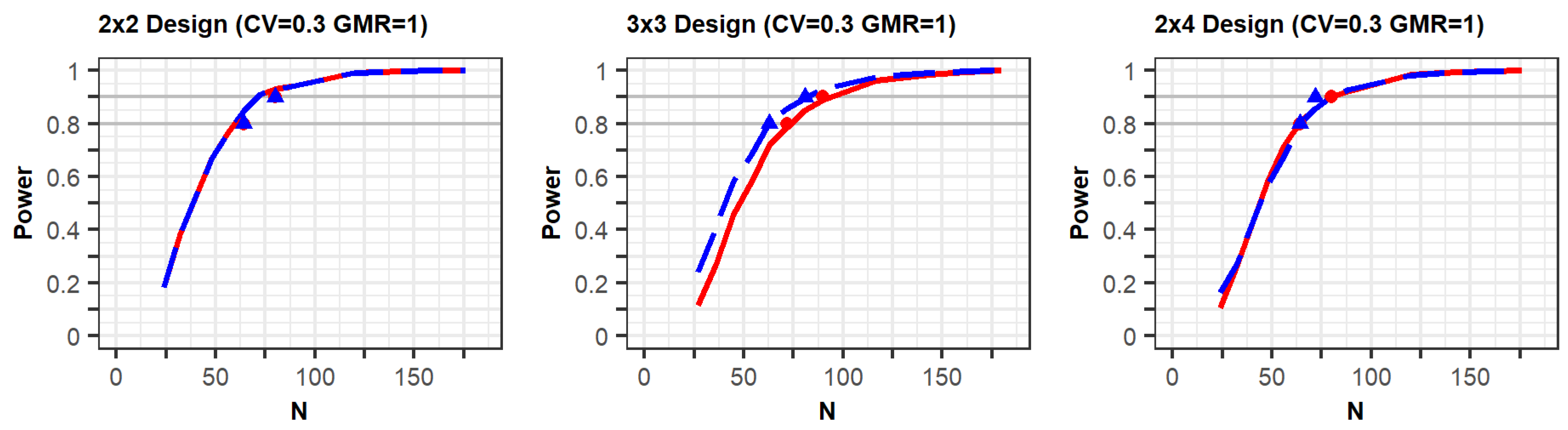
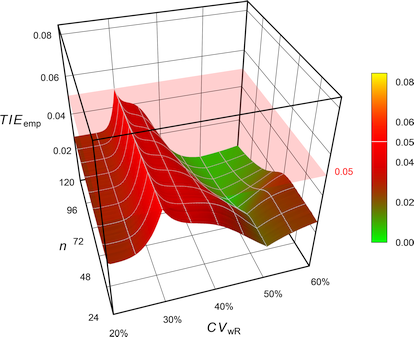
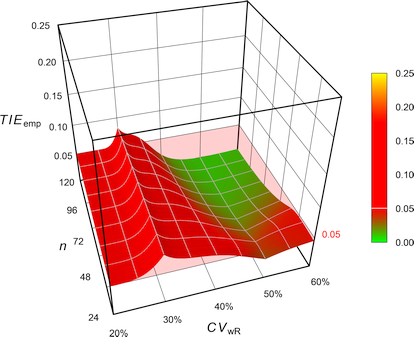
Pharmaceuticals | Free Full-Text | Model-Based Approach for Designing an Efficient Bioequivalence Study for Highly Variable Drugs
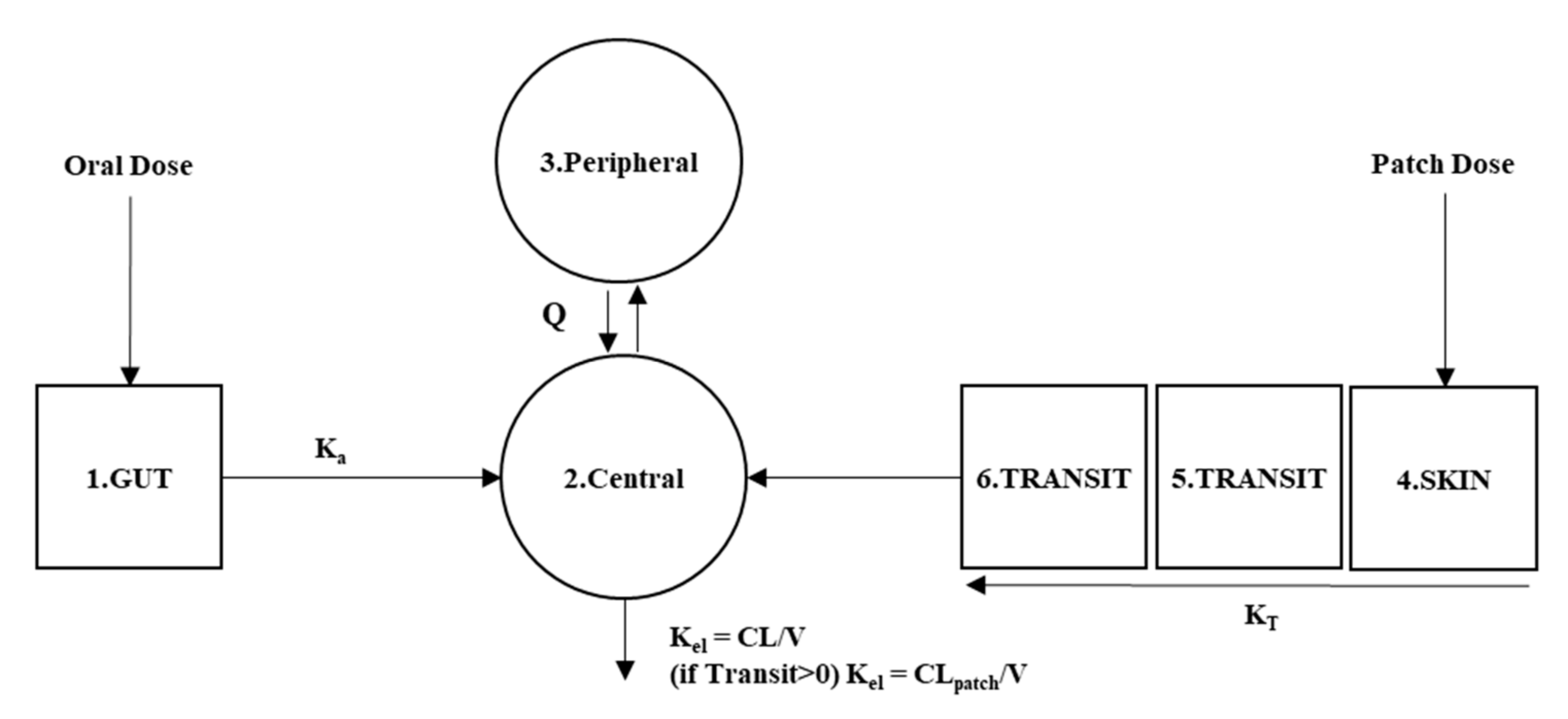
Pharmaceutics | Free Full-Text | Model-Based Equivalent Dose Optimization to Develop New Donepezil Patch Formulation
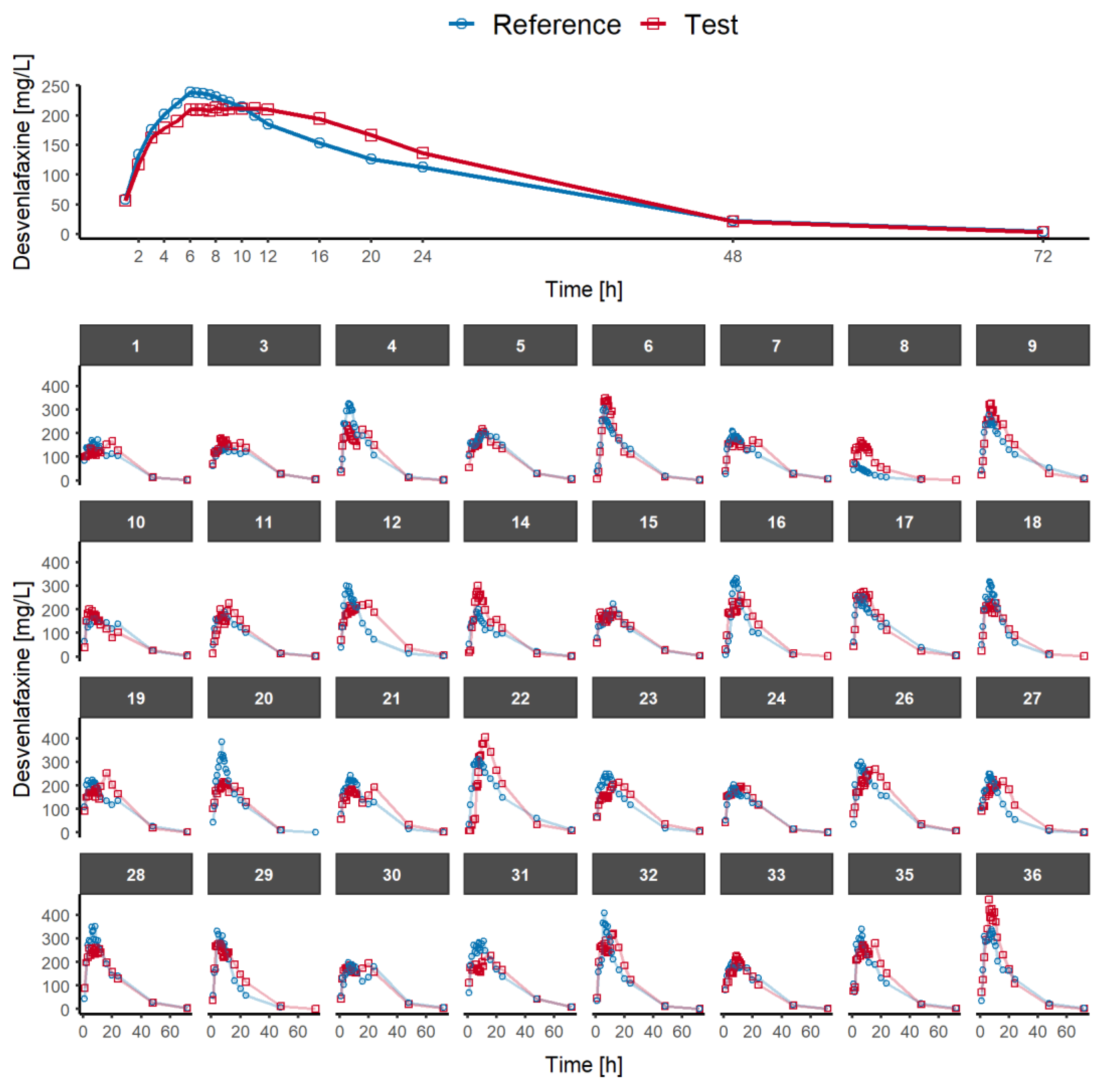
Pharmaceutics | Free Full-Text | Alternative Pharmacokinetic Metrics in Single-Dose Studies to Ensure Bioequivalence of Prolonged-Release Products at Steady State—A Case Study

90% confidence Intervals and point estimates for the ratio of the test... | Download Scientific Diagram

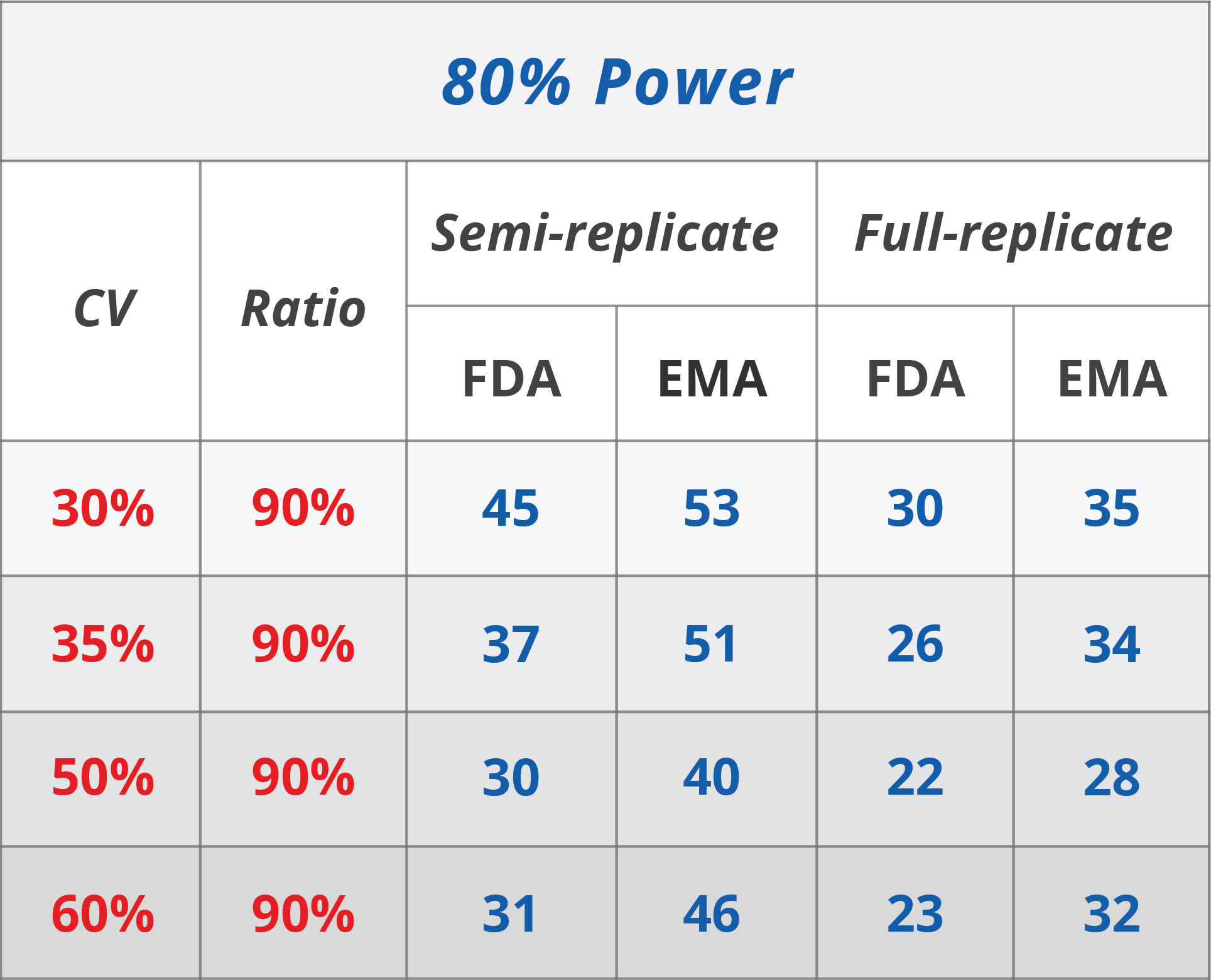
Sample size determination in bioequivalence studies using statistical assurance - Ring - 2019 - British Journal of Clinical Pharmacology - Wiley Online Library

A visual representation of some possible results of the statistical... | Download Scientific Diagram