Simplified flow sheet of nitric acid process showing possible locations... | Download Scientific Diagram
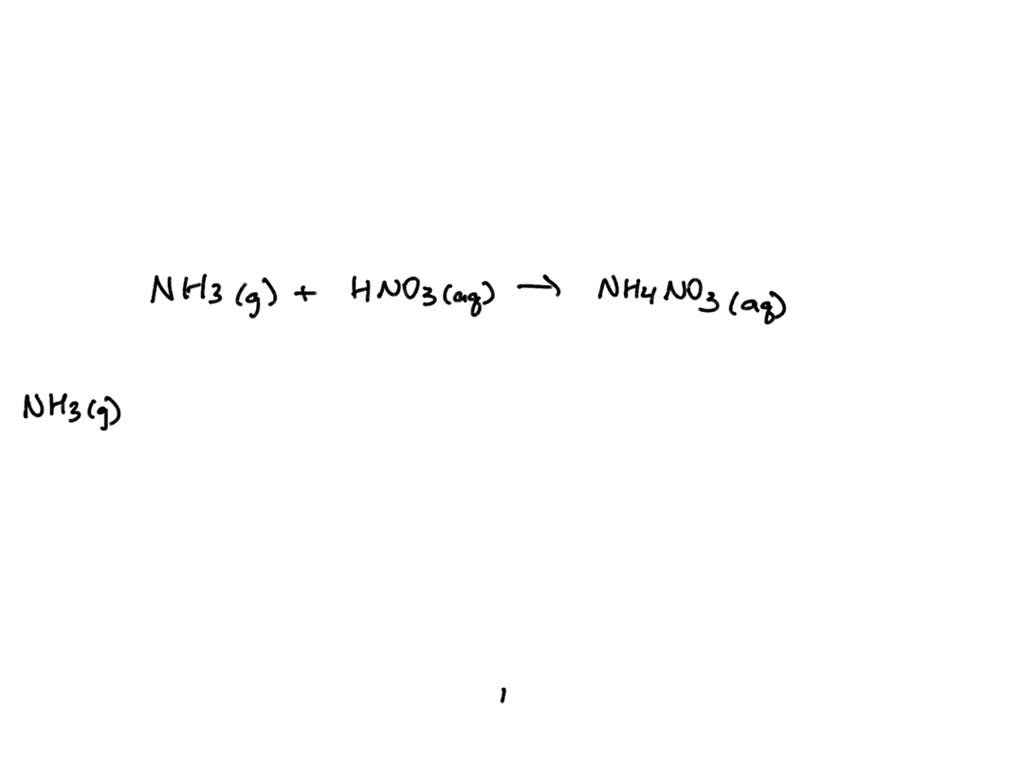
SOLVED: Ammonia gas reacts with aqueous nitric acid to form aqueous ammonium nitrate, a fertilizer. (a) Write the balanced chemical equation for the reaction, including the physical states. (b) Write the complete

Process Intensification in Nitric Acid Plants by Catalytic Oxidation of Nitric Oxide | Industrial & Engineering Chemistry Research

The chloride-catalyzed decomposition of ammonium nitrate in nitric acid media at 100°C - ScienceDirect

Reacting Masses No. moles = mass M r n = m M r. What mass of ammonium nitrate fertiliser can be made from 18.9 g of nitric acid? NH 3 + HNO 3 → NH 4 NO. - ppt download
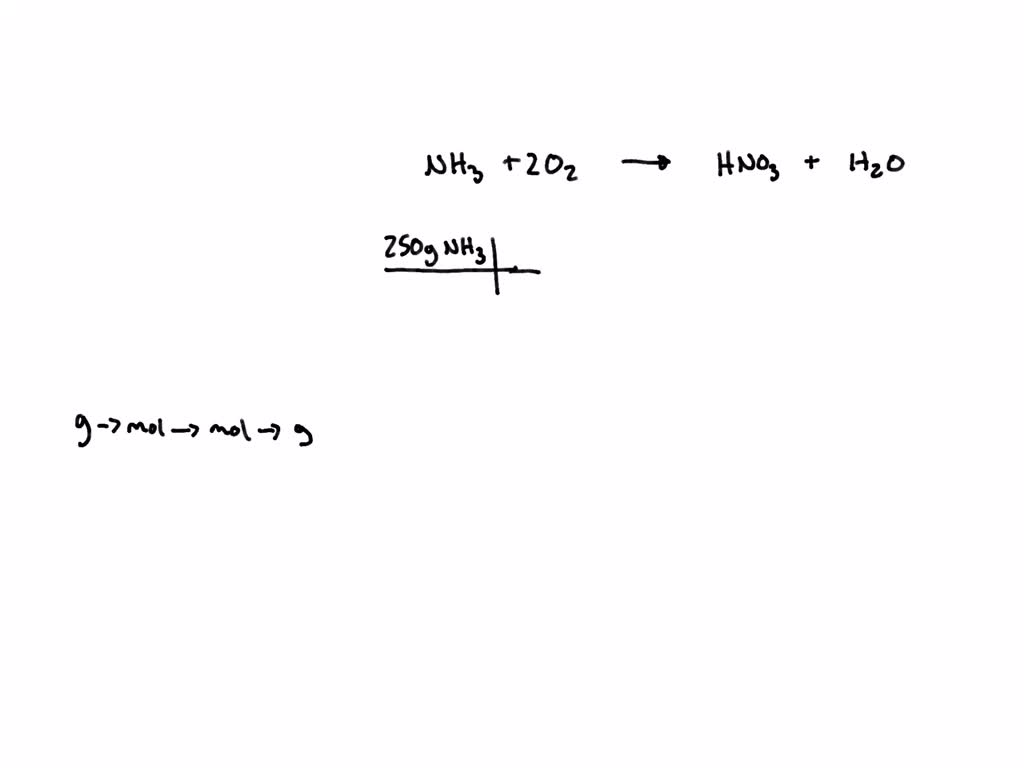
SOLVED: Ammonia reacts with oxygen to produce nitric acid (HNO3) and water. What mass of nitric acid, in grams, can be made from 250 g of ammonia?

SOLVED:The fertilizer, ammonium nitrate, is made by reacting ammonia with nitric acid. (Section 1.4) (a) Write a balanced equation, with state symbols, for the reaction of ammonia gas with nitric acid to

Equilibrium fraction of total ammonia, nitric acid, and hydrochloric... | Download Scientific Diagram

In Ostwald's process for the manufacture of nitric acid, the first step involves the oxidation of ammonia gas by oxygen gas to give nitric oxide gas and steam. What is the maximum

Uses of ammonia nitric acid ammonium nitrate salts fertilisers preparation pollution eutrophication gcse igcse KS4 science chemistry O level revision notes revising

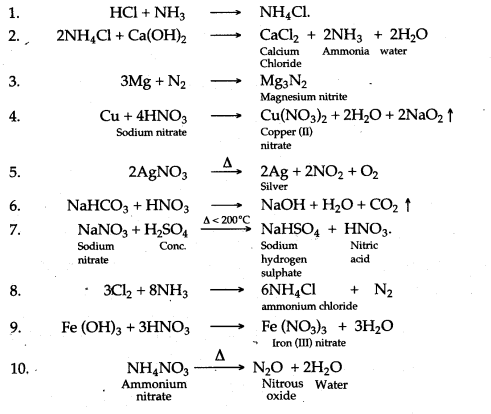
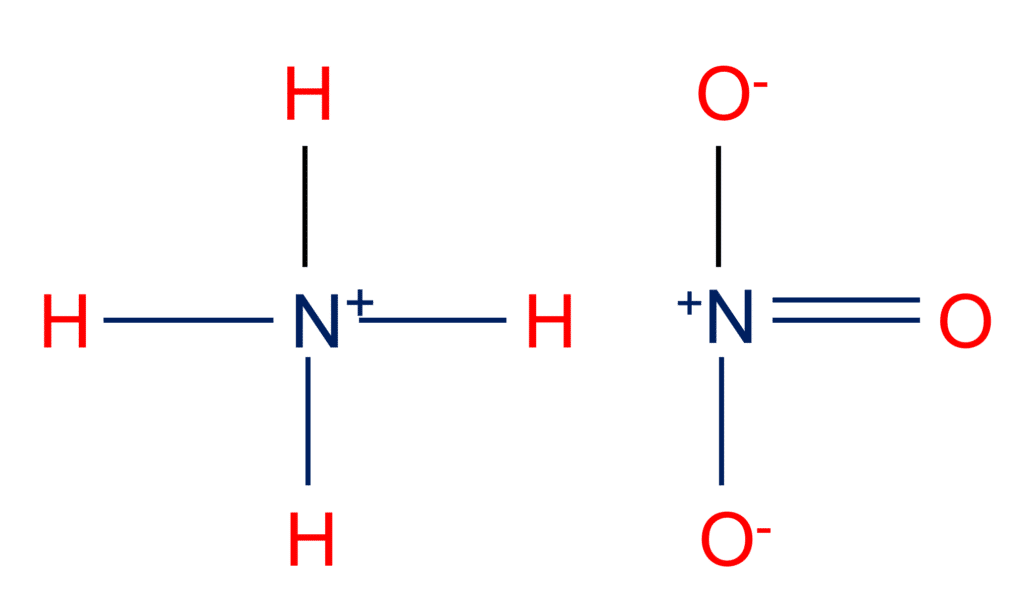


.jpg)





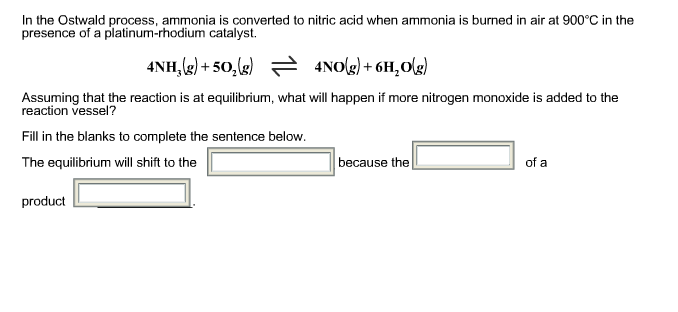


.jpg)

