Write the balanced chemical equation for the following reaction and identify the type of reaction and define - Brainly.in

when a mixture of aluminium powder and iron (III) oxide is ignited, it produces molten iron and - YouTube

Thermit reaction, iron (III) oxide reacts with aluminium andgives molten iron and aluminium oxide. - YouTube

An extensive study on the synthesis of iron based magnetic aluminium oxide nanocomposites by solution combustion method - ScienceDirect

Corundum Is A Crystalline Form Of Aluminium Oxide Typically Containing Traces Of Iron, Titanium, Vanadium. It Is A Rock-forming Mineral. 3d Illustration Stock Photo, Picture And Royalty Free Image. Image 60744188.

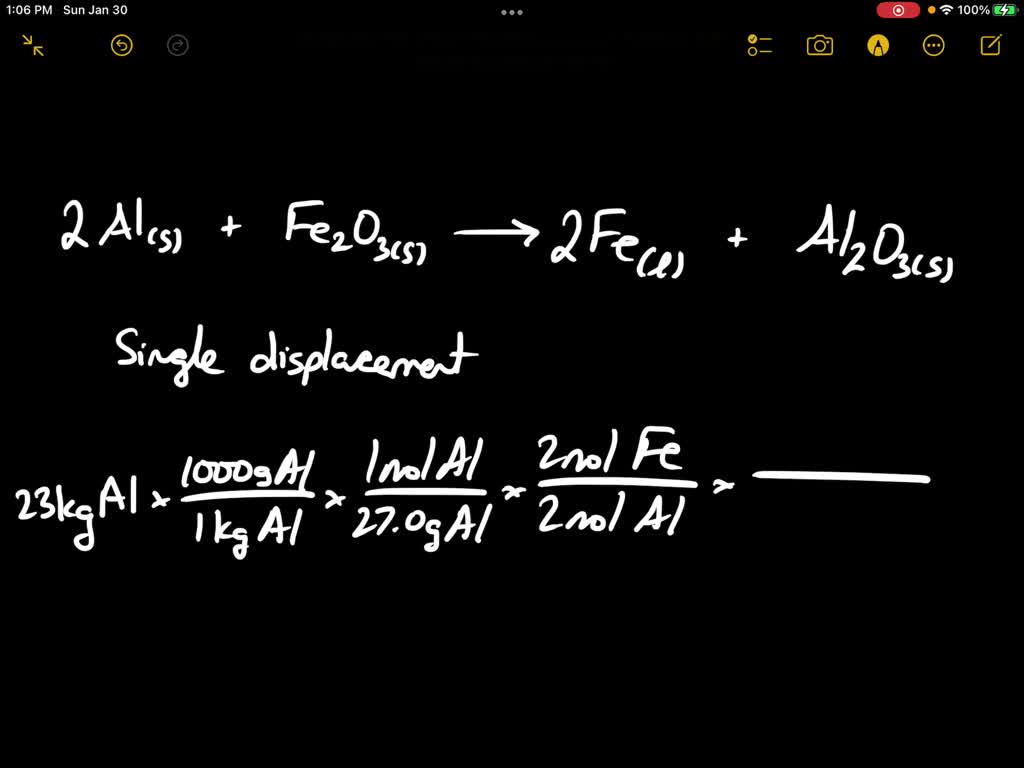
Write the balanced chemical equation for the following reaction and identify the type of reaction and define it. 'Iron III oxide reacts with Aluminium and gives molten iron and aluminium oxide'.

XRD pattern of iron aluminum oxide nanoparticles for oxidation time 5... | Download Scientific Diagram