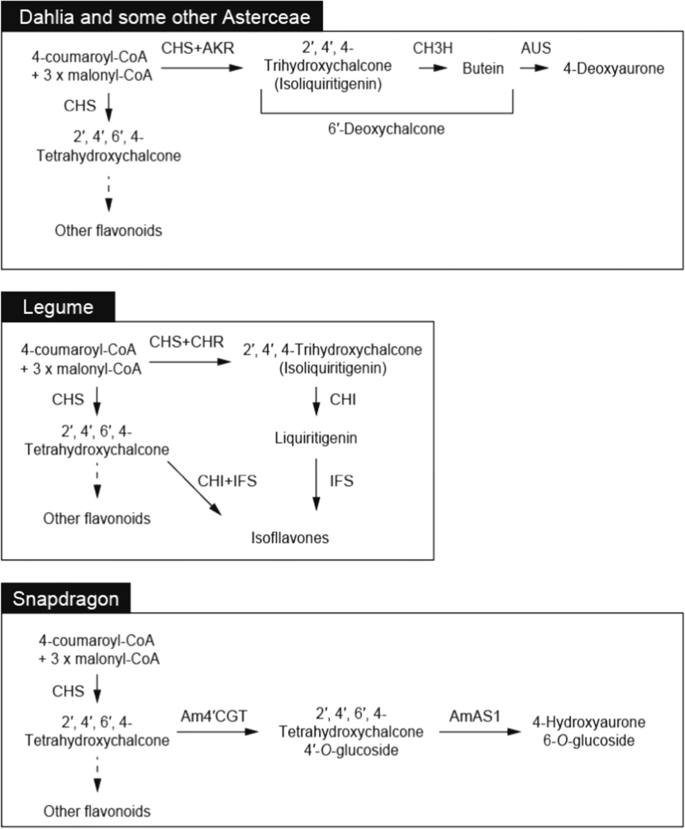
A novel aldo–keto reductase gene is involved in 6′-deoxychalcone biosynthesis in dahlia (Dahlia variabilis) | SpringerLink

Aldo-Keto Reductase Regulation by the Nrf2 System: Implications for Stress Response, Chemotherapy Drug Resistance, and Carcinogenesis. - Abstract - Europe PMC
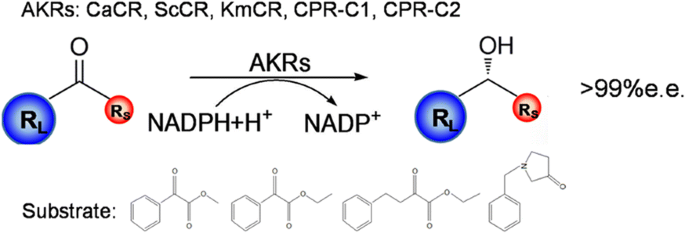
Gene mining-based identification of aldo–keto reductases for highly stereoselective reduction of bulky ketones | Bioresources and Bioprocessing | Full Text
![PDF] Inhibitor selectivity between aldo – keto reductase superfamily members AKR 1 B 10 and AKR 1 B 1 : Role of Trp 112 ( Trp 111 ) | Semantic Scholar PDF] Inhibitor selectivity between aldo – keto reductase superfamily members AKR 1 B 10 and AKR 1 B 1 : Role of Trp 112 ( Trp 111 ) | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/d7db25f0752a33fda3a5d5d8f1ff49b98a97bd23/5-Figure3-1.png)
PDF] Inhibitor selectivity between aldo – keto reductase superfamily members AKR 1 B 10 and AKR 1 B 1 : Role of Trp 112 ( Trp 111 ) | Semantic Scholar
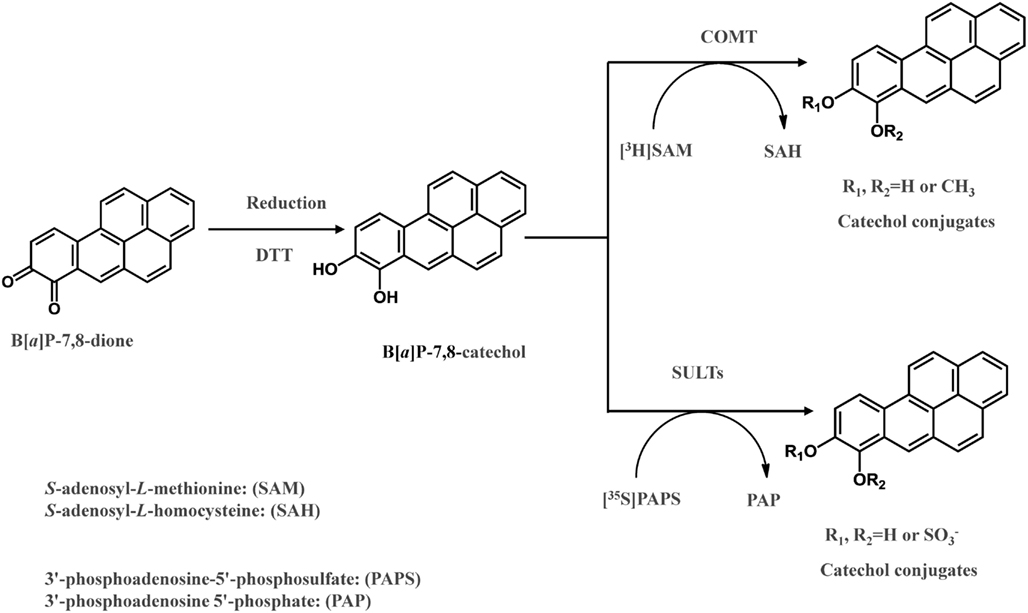
Frontiers | The Role of Human Aldo-Keto Reductases in the Metabolic Activation and Detoxication of Polycyclic Aromatic Hydrocarbons: Interconversion of PAH Catechols and PAH o-Quinones
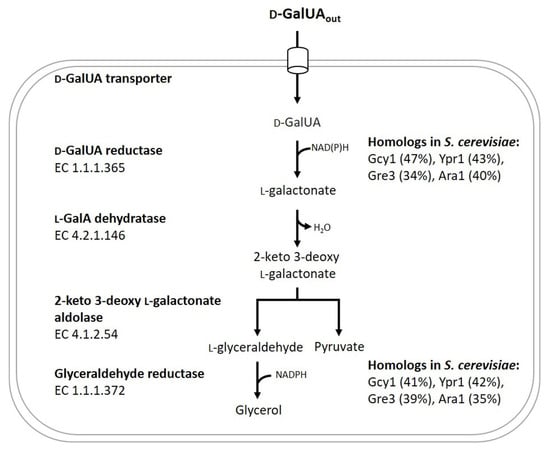
JoF | Free Full-Text | Identification of the Aldo-Keto Reductase Responsible for d-Galacturonic Acid Conversion to l-Galactonate in Saccharomyces cerevisiae

Structure–function characterization of an aldo–keto reductase involved in detoxification of the mycotoxin, deoxynivalenol | Scientific Reports

Discovery of Novel Aldo-Keto Reductase 1C3 Inhibitors as Chemotherapeutic Potentiators for Cancer Drug Resistance | ACS Medicinal Chemistry Letters

Human Aldo-Keto Reductases and the Metabolic Activation of Polycyclic Aromatic Hydrocarbons | Chemical Research in Toxicology

IUCr) Structure–function study of AKR4C14, an aldo-keto reductase from Thai jasmine rice (Oryza sativa L. ssp. indica cv. KDML105)
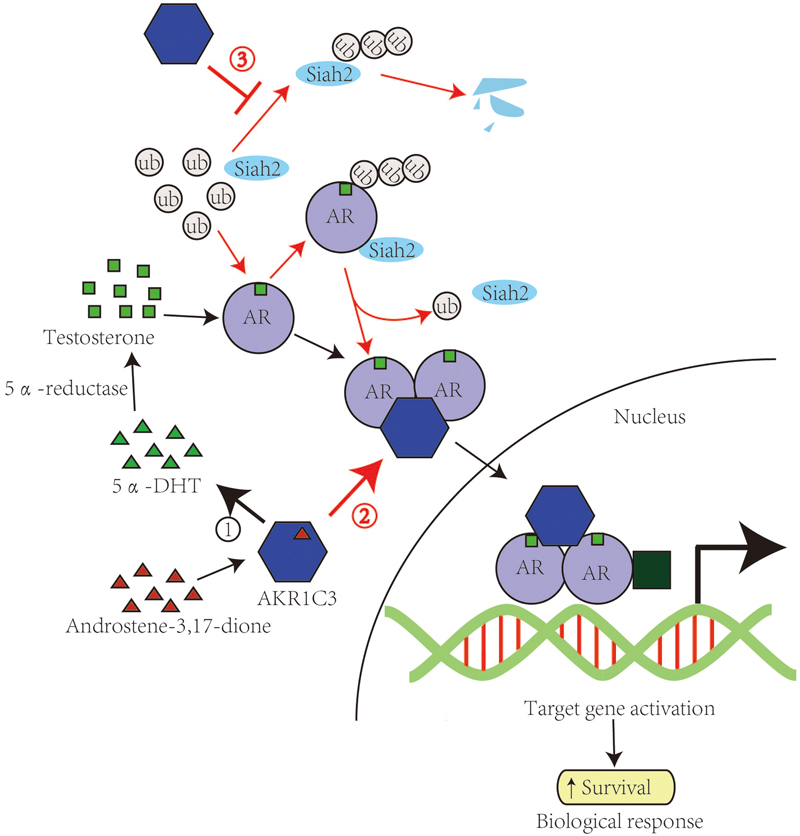
Frontiers | Aldo–Keto Reductase AKR1C1–AKR1C4: Functions, Regulation, and Intervention for Anti-cancer Therapy












