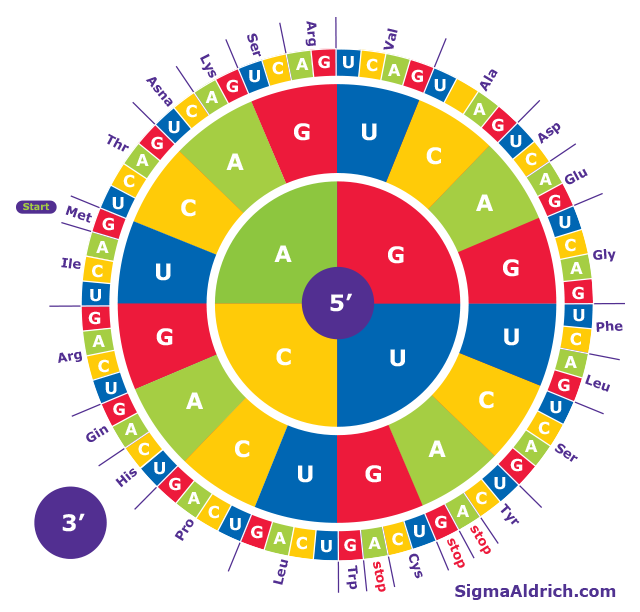
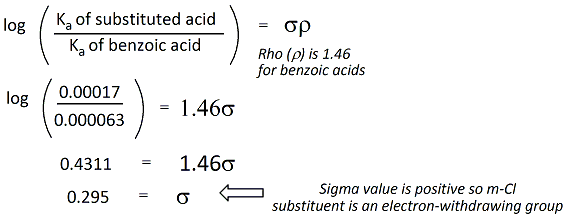TI-89 Calculator - 01 - Taking Derivatives of a Function in Calculus - TI-89 Calculator - Vol 2 - Math Tutor Public Gallery
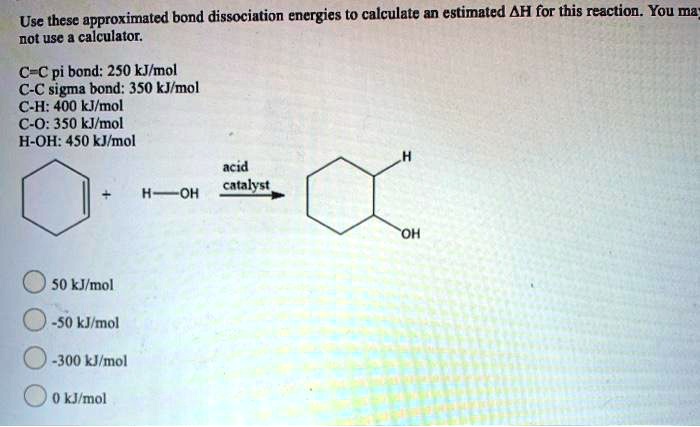
SOLVED: Use these approximated bond dissociation energies to calculate an estimated AH for this reaction. You ma= not use calculator: C-Cpi bond: 250 kJlmol CC sigma bond: 350 kJmol C-H: 400 kJlmol
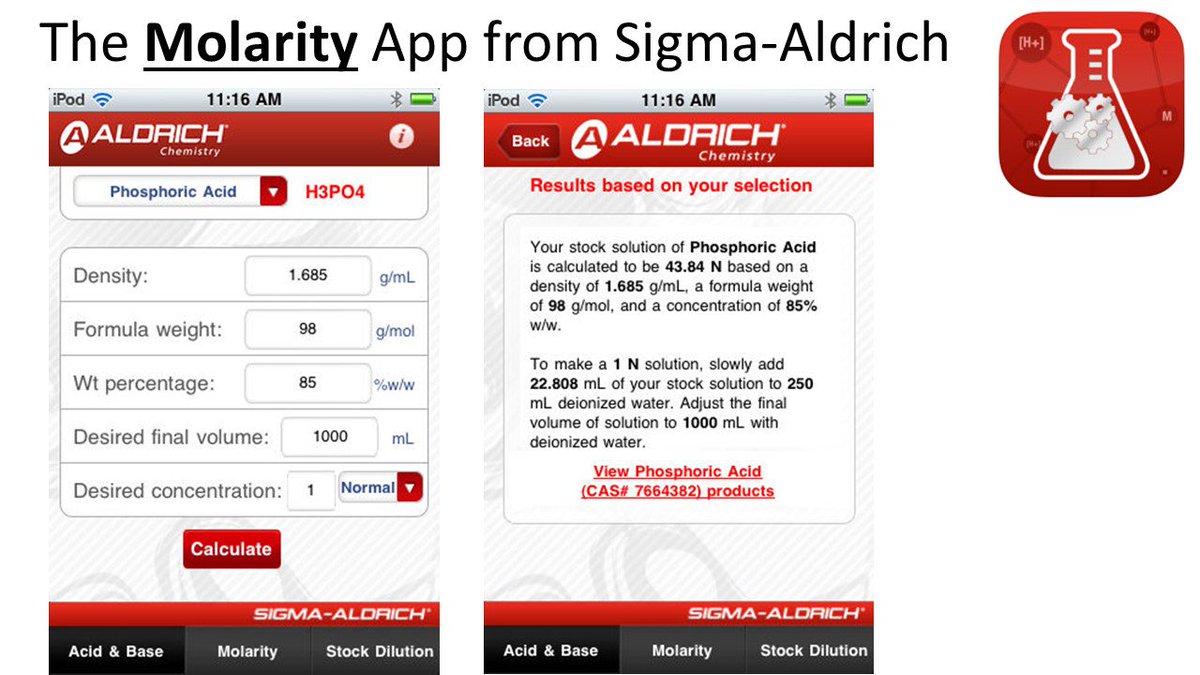
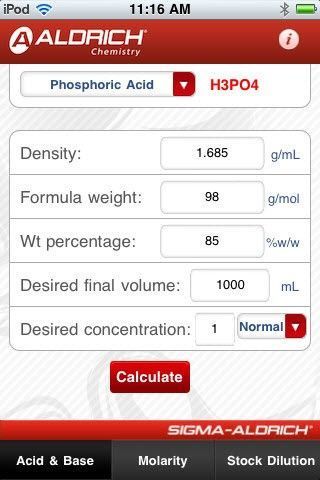
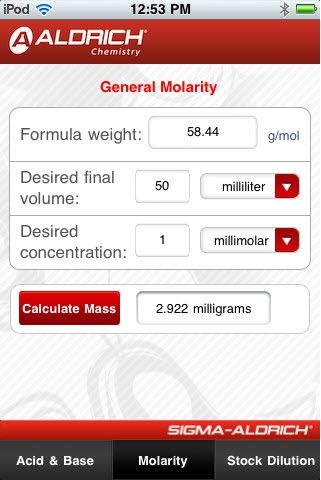
Ellen Hackl 🇺🇦 🇬🇧 on Twitter: "Do you need to prepare 0.2 N nitric acid? You can use the Normality & Molarity Calculator from Sigma-Aldrich. https://t.co/Mr2IHvclAc Or download the Molarity App for
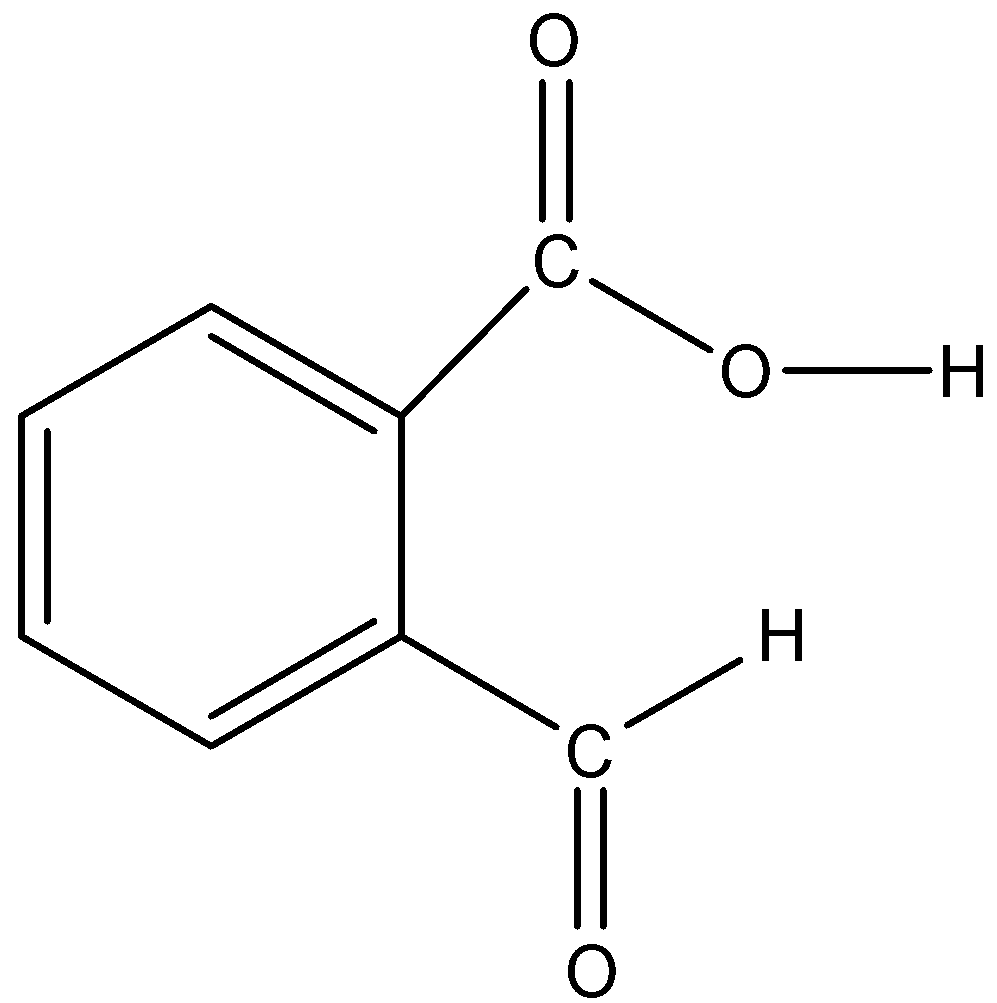


-Malic%20acid[800763_D-(+)-Malic%20acid-ALL].jpg)