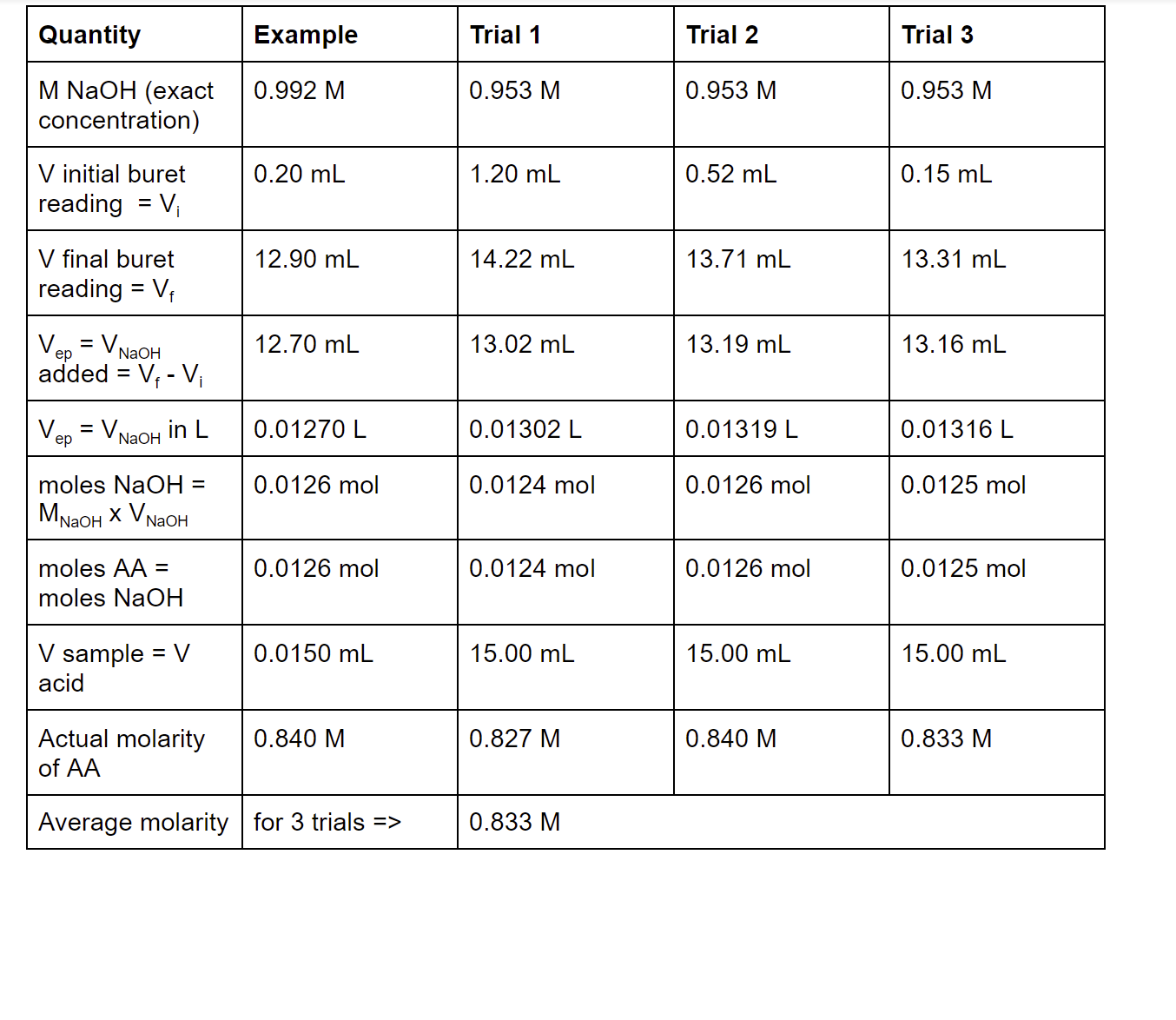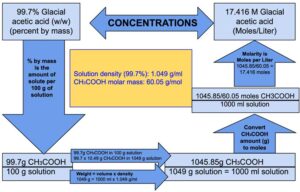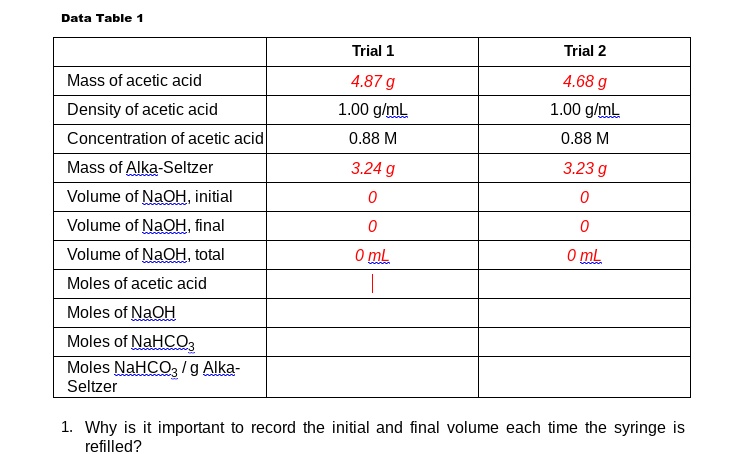
SOLVED: Data Table Trial 1 Trial 2 Mass of acetic acid Density of acetic acid Concentration of acetic acid Mass of Alka-Seltzer Volume of NaQH, initial Volume of NaQH; final Volume of

Effect of chitosan dissolved in different acetic acid concentration towards VOC sensing performance of quartz crystal microbalance overlay with chitosan - ScienceDirect

Influence of acetic acid concentration on pH of chitosan solution and... | Download Scientific Diagram

The dissociation constants for acetic acid and HCN at 25C are 1.5 x 10^-5 and 4.5 x 10^-10 respectively. The equilibrium constant for the equilibrium CN + CH3COOH HCN + CH3COO - would be ?
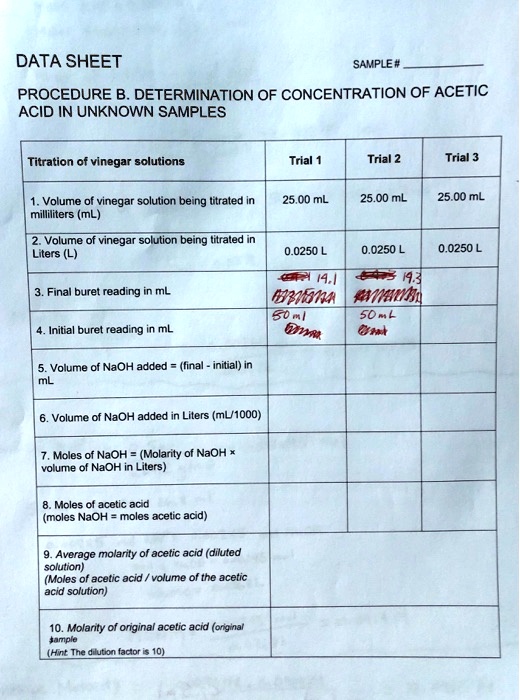
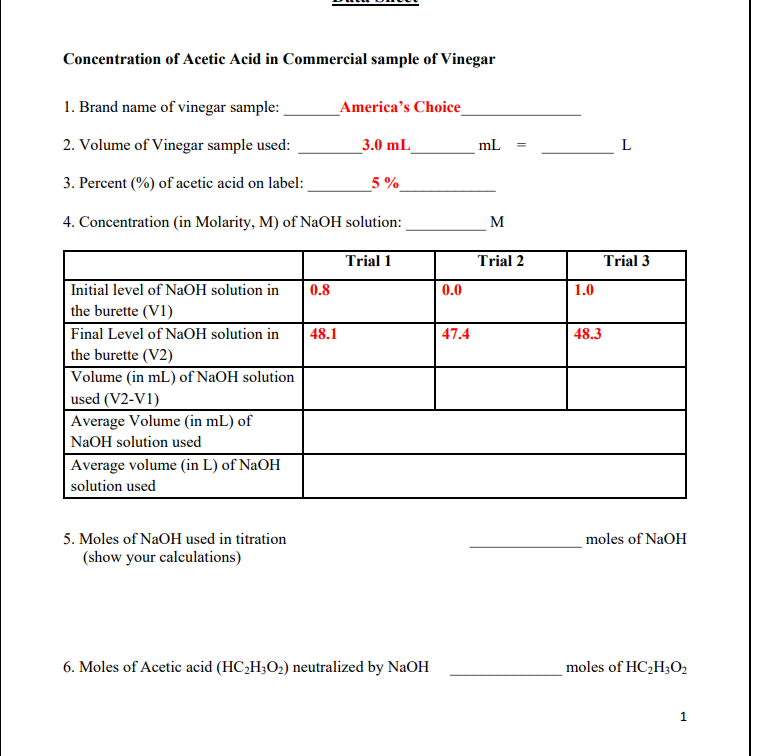
SOLVED: DATA SHEET SAMPLE# PROCEDURE B. DETERMINATION OF CONCENTRATION OF ACETIC ACID IN UNKNOWN SAMPLES Titration of vinegar solutions Trial Trial 2 Trial Volume of vinegar solution being litraled 5 milliliters (ML)
The pH of an acetic acid solution is 3.26. What is the concentration of acetic acid and what is the percent of acid that's ionized? - Quora
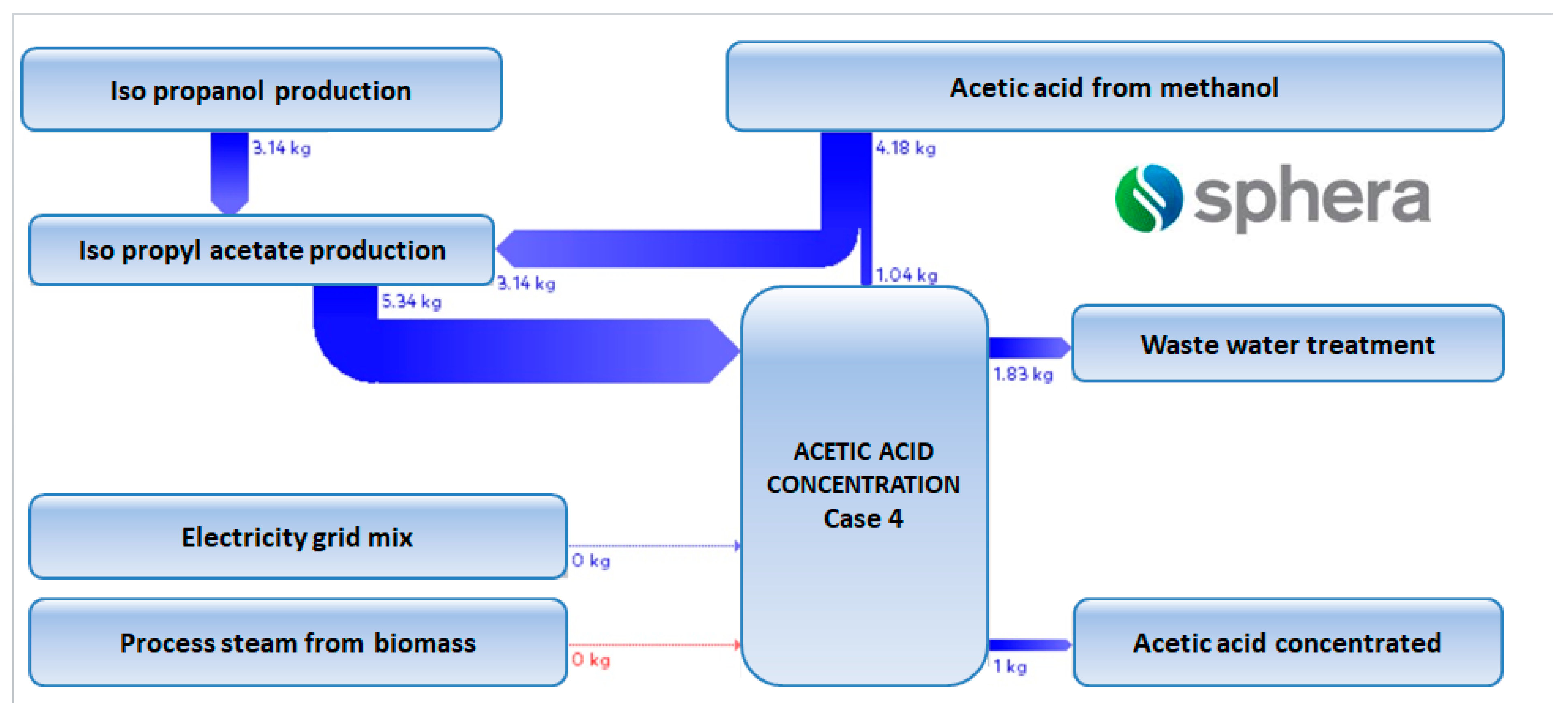
Energies | Free Full-Text | Classical and Process Intensification Methods for Acetic Acid Concentration: Technical and Environmental Assessment