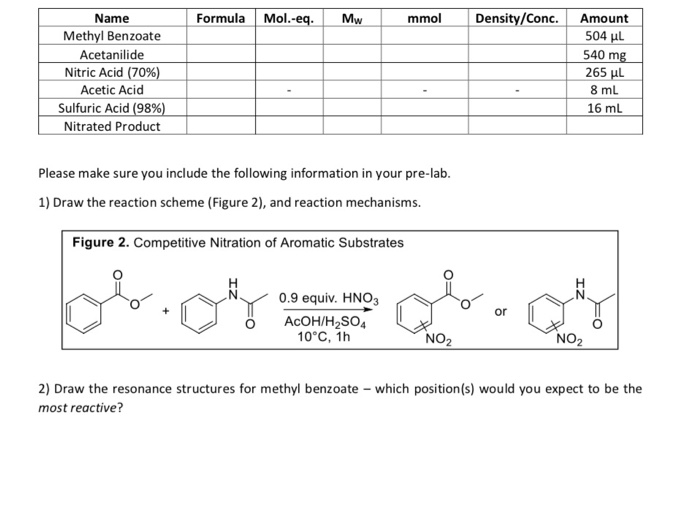
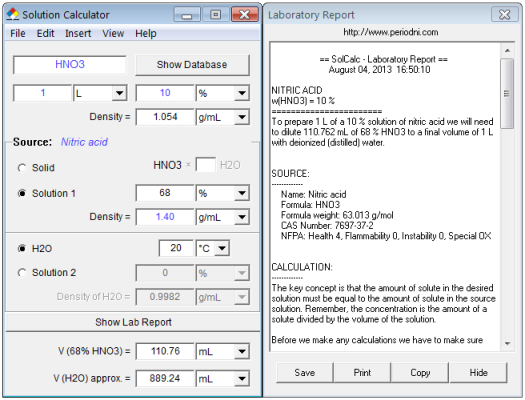
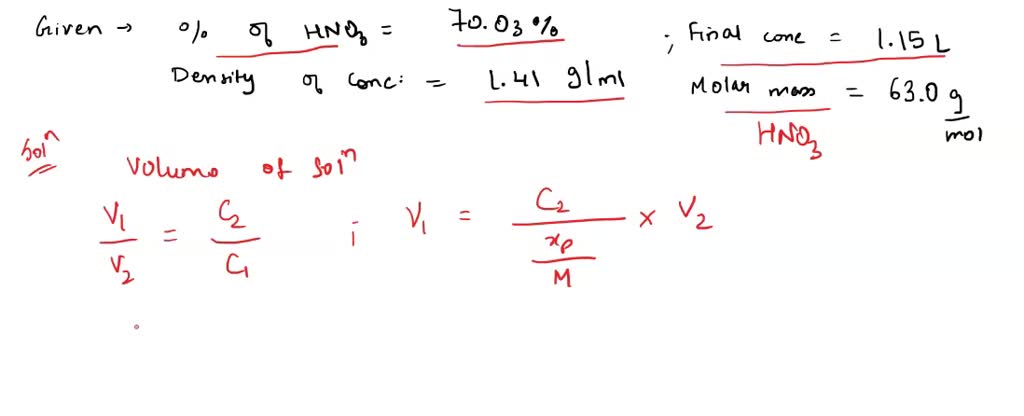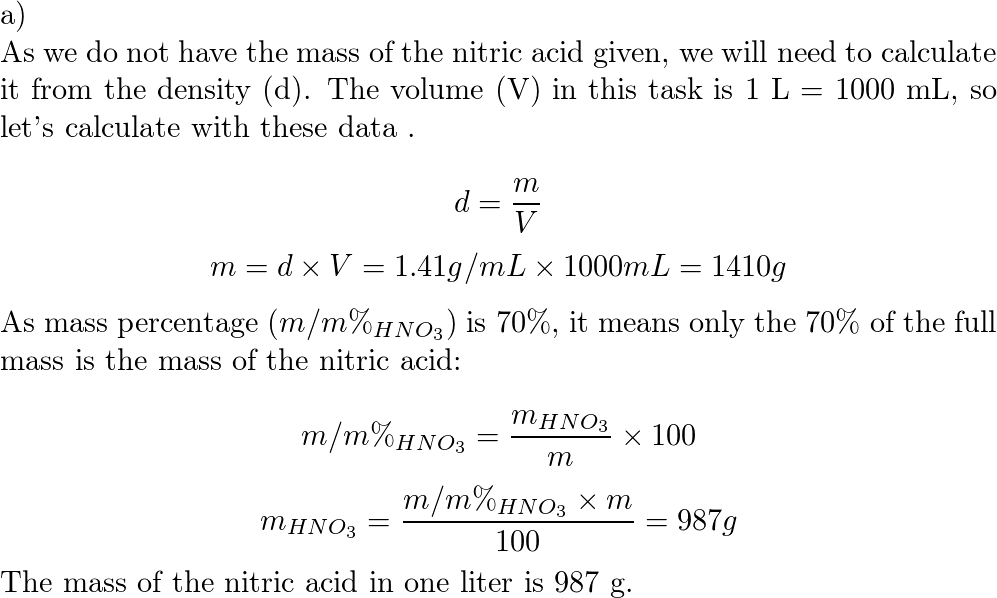How many grams of concentrated nitric acid solution should be used to prepare 250 mL of 2.0 M HNO3 ? The concentrated acid is 70

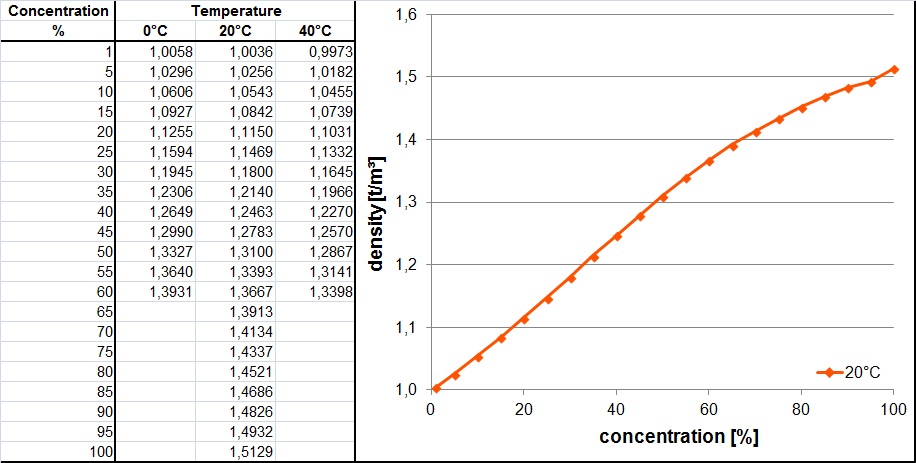
SOLVED: Nitric Acid is listed as 70% wt Wt It has a density of 1.413 mL #Hint: start with a liter of solution and use the density to calculate the mass of

SOLVED: a solution is prepared with 70.0 g of nitroc acid and 130.0 g of water. the nitric acid solution had a density of 1.21 g/ml a) what is he mass percent